GM TransExcellent Plasmid DNA Rapid Preparation Service
--1 week, higher efficiency, good for your proteins, antibodies, and viral vector production
GeneMedi offers a plasmid production&lification service called GM TransExcellent Plasmid DNA Rapid Preparation Service for DNA vector preparation based on the G-NEXT™ platform, which is a fast (1 week), high-quality, high-efficiency, and cost-effective plasmid production&lification platform and is good for your proteins, antibodies, and viral vector production.
GeneMedi ensures the quality of the final products through strict adherence to standard operating procedures, perfect quality management, strict testing systems, and traceable operating procedures. The platform's large-scale industrial fermentation and fully automated purification process production can accommodate the service needs of industrial users for large-scale plasmid preparation. Furthermore, GeneMedi's Novel Evolution-X Technology (G-NEXT™) is used to explore Gene&Cell therapy (GCT) for more possibilities with innovational strategies.
- How to order
- Validation data
- Plasmid source
- Quality Levels
- Plasmid preparation period
- Production steps
How to order
To place an order for GM TransExcellent Plasmid DNA Rapid Preparation Service, please follow the steps below:
STEP 1: Confirm the necessary information as follows:
a. Confirm the plasmid:
If you are using/will use GENEMEDI's vector system product, please provide us with the Catalog No.(Click here to check the Cat No.)
If you want to use your own plasmid, please provide us with the sequence. After placing your order, please send us 2ug of plasmid.
b. Confirm the Quality Levels of Plasmid DNA:
choose from Research Grade, Endotoxin-Free Grade or High Quality (HQ) Grade by checking out the detail of different levels.
c. Confirm the Plasmid quantity:
choose 10mg or more by checking the period of different quantity.
STEP 2: Submit the confirmed information using the format below:
Cat No.: P-RC13
Quality Level: Research Grade
Plasmid quantity: 10mg
Submit
Submit Example:
You can also send the required information via email to ([email protected]).
Our team will process your request within 1 business day and guide you through the ordering process.
Terms of sale:
The purchaser may not reverse engineer this product to extract the sequence for independent use. The purchaser may not transfer this product to others for manufacturing purposes. The purchaser may not use this material to manufacture this product for themselves or any other party.
Validation data
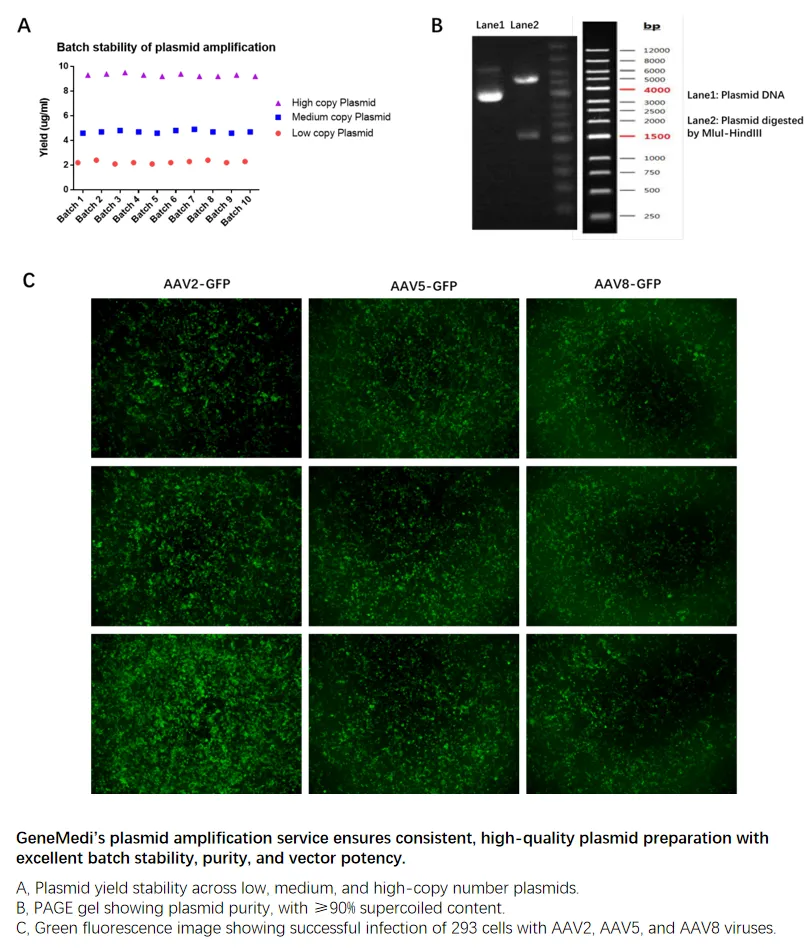
GeneMedi’s plasmid amplification service ensures consistent, high-quality plasmid preparation with excellent batch stability, purity, and vector potency
1. Consistent Batch-to-Batch Yield Stability
As shown in Figure A, GeneMedi’s plasmid amplification service delivers stable yields across different batches, whether for low, medium, or high-copy plasmids. This consistency ensures reliable results for varied applications, demonstrating the robustness of the service.
2. High Purity Plasmids with ≥90% Supercoiled Content
Figure B presents the PAGE gel, confirming that GeneMedi's amplified plasmids exhibit high purity, with ≥90% supercoiled content. This purity level guarantees that the plasmids are of the highest quality for demanding research or therapeutic applications.
3. Excellent Vector Potency for Efficient Gene Delivery
Figure C shows the green fluorescence from 293 cells infected with AAV2, AAV5, and AAV8 viruses, demonstrating the enhanced potency of the AAV vectors prepared using GeneMedi’s plasmids. This high infectivity ensures effective gene delivery for gene therapy research.
Fig. GeneMedi’s plasmid amplification service ensures consistent, high-quality plasmid preparation with excellent batch stability, purity, and vector potency.
A, Plasmid yield stability across low, medium, and high-copy number plasmids.
B, PAGE gel showing plasmid purity, with ≥90% supercoiled content.
C, Green fluorescence image showing successful infection of 293 cells with AAV2, AAV5, and AAV8 viruses.
Plasmid source: GeneMedi's vector system products & Your own plasmid
1.GeneMedi's vector system products includes:
| Products | Details |
| AAV vector system (AAV expression system, AAV packaging plasmid system) |
Serotype: AAV1, AAV2, AAV2, AAV2 variant (Y444F), AAV2 variant (Y272F, Y444F, Y500F, Y730F), AAV2 variant (Y444F, Y730F, Y500F, Y272F, Y704F, Y252F), AAV2.7m8, AAV5, AAV6, AAV8, AAV8-1m, AAV8-2m, AAV8-3m, AAV9, AAV-Rh10, AAV-DJ, AAV-DJ8, AAV2-Retro(Retrograde), AAV-PHP.B, AAV-PHP.eB, AAV-PHP.S, AAV-BR1, AAV-VEC, AAV-2i8, AAV-SIG, AAV4, AAV6.2, AAV6.2FF, MyoAAV 1A, MyoAAV 2A, MyoAAV 3A, MyoAAV 4A, MyoAAV 4E, AAV-Anc80, AAV-rh74. AAV-LK03 |
| Adenovirus expression vectors (adenovirus expression plasmids) | |
| Lentivirus vector system | Including PMD2G(Cat No.: P-ALV-C01), pSPAX2(Cat No.: P-ALV-C02) and other expression vector |
| Promise-ORF™ viral CDNA library | More than 60,000 pre-made CDNA/ORF viral plasmids and viral particles in Lentivirus, Adenovirus and AAV |
2.Your own plasmid
Quality Levels of Plasmid DNA
GeneMedi provides three types of Quality Levels of Plasmid DNA: Research Grade, Endotoxin-Free Grade, and High Quality (HQ) Grade. The provided plasmids are compared in Table 1.
| QC Item | Service Grades | ||
| Research Grade | Endotoxin-Free Grade | High Quality (HQ) Grade | |
| E.coli Master Cell Bank generation | - | - | Optional |
| Manufacturing via shake flask growth or high-density fermentation tank | shake flask growth or high-density fermentation tank | shake flask growth or high-density fermentation tank | high-density fermentation tank |
| Screening for optimal growth conditions | - | - | √ |
| Alkaline lysis | √ | √ | √ |
| Purification method | Anion exchange chromatography | Anion exchange chromatography, Specialized removal of endotoxins | Gel filtration chromatography, Affinity chromatography, Anion exchange chromatography,Specialized removal of endotoxins |
| Appearance | √ | √ | √ |
| pH | - | - | √ |
| A260:280 | √ | √ | √ |
| Residual RNA | Non-detectable by gel electrophoresis at 200ng | ||
| Residual E.coli DNA | Non-detectable by gel electrophoresis | Quantitative PCR ≤5% | |
| Residual Host Protein | - | - | HCP ELISA ≤ 1% |
| Homogeneity | Predominantly supercoiled | ≥90% supercoiled | |
| Animal Free Production | Optional | Optional | Optional |
| Enzyme Free production | - | - | Optional |
| Restriction Analysis | - | √ | √ |
| Identity-Full Insert Sequencing | √ | √ | √ |
| Endotoxin | - | Gel clot LAL assay < 10EU/mg | Quantitative LAL assay < 5EU/mg |
| Bioburden Testing | √ | √ | √ |
| Mycoplasma Contamination | - | - | Optional |
| Detection of Kanamycin | - | - | Optional |
| Material Archiving | - | - | Optional |
| TSE/BSE Certificate | - | - | √ |
| Certificate of Analysis | √ | √ | √ |
| Consistent Manufacturing Process | √ | √ | √ |
| Quality oversight | - | - | √ |
Table 1. Comparation of different grades of Plasmids GeneMedi provides.
Plasmid preparation period
GeneMedi has a fast delivery period advantage, and below are the Preparation periods of different grades of Plasmids provided in Table 2.
| Plasmid Grade | Plasmid quantity | Period (working days) |
| Research Grade | 10mg | 5~10 | 50mg | 7~12 | 100mg | 10~15 | 500mg | 15~20 | ≥1g | Inquiry | Other quantity | Inquiry |
| Endotoxin-Free Grade | 10mg | 5~10 | 50mg | 10~15 | 100mg | 15~20 | 500mg | 20~25 | ≥1g | Inquiry | Other quantity | Inquiry |
| High Quality (HQ) Grade | Inquiry | Inquiry |
Table 2. Preparation periods of different grades of Plasmids GeneMedi provides.
Production steps of Plasmids
Large-scale plasmid DNA manufacturing includes five main steps, as shown in Figure 1.
1. E. coli cells used for plasmid DNA manufacturing were obtained from sources such as DSMZ (German Collection of Microorganisms and Cell Cultures). The transformed cell clones are tested for plasmid identity by restriction analysis, and—if requested—further analysis is applied.
2. A pre-culture of approx. 50 mL is used to inoculate the main culture of 30 L. Cells are cultivated overnight at typically 37 °C or—if necessary to maintain unstable sequences or desired by the customer—at lower temperatures. Cells are harvested on this scale by batch centrifugation.
3. Alkaline lysis is performed by a rapid alka_x0002_line extraction procedure, as published earlier. Significant modification is necessary to scale up this step, as discussed later. Removal of residual particles, cells, or cell fragments is performed by batch centrifugation and multiple filtration steps, including a final 0.2 μm filtration of the “cleared lysate”.
4. Chromatographic steps are performed to reduce the volume (chromatography) and to remove (additional chromatography) contaminating LPS endotoxins, RNA, proteins, and undesired DNA contaminants (chromosomal bacterial DNA, non-supercoiled plasmid forms). The final eluate is filtered (0.2 μm), precipitated, washed, and reconstituted within the buffer.
5. According to the requested aliquots, certified cryotubes are manually filled within a laminar airflow. Vials are labeled and stored under quarantine until the product is released after QC at approx. −20 °C.
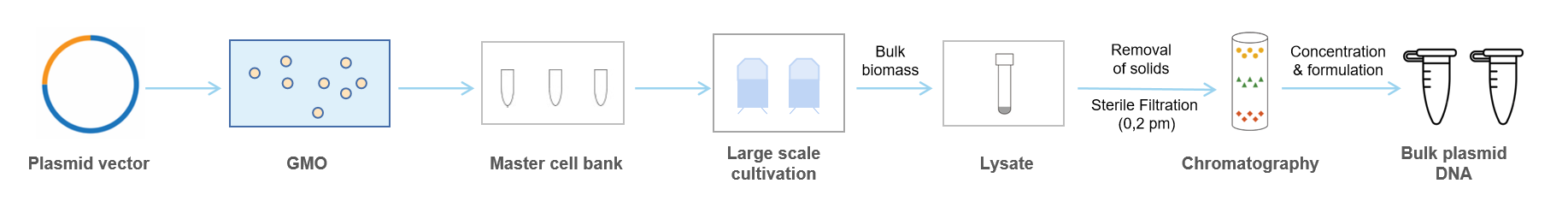
Figure 1. Production steps in large-scale plasmid DNA manufacturing from plasmid DNA construct to final product.




