Immuno-Diagnostics Antibodies and Antigens for Food/Feed Safety and Environmental Analysis
GeneMedi's product list for Food/Feed Safety and Environmental Analysis
Category
About Food/Feed Safety and Environmental Analysis
Competitive immunoassay-validated anti-Hapten(small molecules, chemicals) antibody against hapten (small molecules, chemicals) and hapten-carrier conjugated competitive antigens(Carrier-coupled antigen,immunogen,hapten-carrier conjugates, BSA-conjugated, OVA-conjugated).
The anti-Hapten antibodies against haptens had been validated with our hapten-carrier conjugates via competitive ELISA test.
GeneMedi offers paired anti-small molecules (Chemicals, Antibiotics,Mycotoxins,Hormones,Drugs of Abuse, etc.) antibodies (monoclonal antibody, mab) and small molecules competitive antigens (Carrier-coupled antigen, immunogen, hapten-carrier conjugates, BSA-conjugated, OVA-conjugated) for the rapid test and diagnostics kit of Growth Promoters, Toxic Heavy Metal, Myotoxins,Nutritions, Food Safety, Pesticides, Drugs of Abuse, Allergen, Antibiotics, Agricultural, Water Contamination and so on.
All our anti-chemicals antibodies and small molecules hapten-carrier conjugates antigens (BSA-conjugated, OVA-conjugated)are suitable for in competitive ELISA, Lateral flow immunoassay (LFIA) and other immunoassays in diagnostics and rapid test kit. The carrier-coupled antigens of small molecules can act as immunogens.
Validation Data
Trimethoprim is a synthetic derivative of trimethoxybenzyl-pyrimidine with antibacterial and antiprotozoal properties. Sulfamethoxazole with trimethoprim is a fixed antibiotic combination that is widely used for mild-to-moderate bacterial infections and as prophylaxis against opportunistic infections. Recently, our R&D department demonstrated that our GMP-SMT-73-Ab-1 (Anti-Trimethoprim(TMP) mouse monoclonal antibody) has a large linear range and good sensitivity against the GMP-SMT-73-Ag-1 (BSA-Trimethoprim(TMP)). Below is the result of GeneMedi's Anti-Trimethoprim(TMP) mouse monoclonal antibody validation with BSA-Trimethoprim(TMP) in ELISA. We highly recommend the Ab&Ag to you.
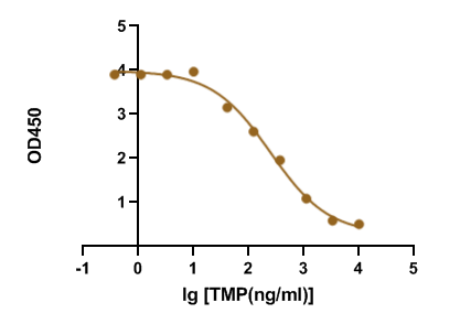
Figure 1. GeneMedi's GMP-SMT-73-Ab-1 (Anti-Trimethoprim(TMP) mouse monoclonal antibody) is validated to detect the GMP-SMT-73-Ag-1 (BSA-Trimethoprim(TMP)) in ELISA. EC50 = 234.5 ng/ml.




 Full List Download
Full List Download
