Bispecific antibodies: formats, applications and products
Products Introduction
Bispecific antibodies (BsAbs) are antibodies with two binding sites directed at two different antigens or two different epitopes on the same antigen. A bispecific antibody (bsAb) is designed and manufactured—through genetic recombination, chemical conjugation, or quadromas—to contain two target‐binding units in one antibody‐based molecule, whereby each unit independently recognizes its unique epitope. The clinical therapeutic effects of BsAbs are superior to those of monoclonal antibodies, with broad applications for tumor immunotherapy as well as for the treatment of other diseases. To meet the needs of BsAbs development, GeneMedi provides more than 500+ BsAbs products.
Case Study -- GeneMedi's GMP-Bios-ab-528 (Pre-Made Solitomab biosimilar) is validated to bind the Jurkat cells by Flow Cytometry (FACS Analysis)
GM R&D department tested the binding ability of GeneMedi's GMP-Bios-ab-528 (Pre-Made Solitomab biosimilar) to the Jurkat cells by Flow Cytometry. Below is the conclusion: By binding with 2 antigens from 2 different cells, the BsAbs can physically link them together, thus these kinds of BsAbs are named “engagers”. The redirecting of immune cells to tumor cells by the engagers makes the immune cell activated and then eliminate the target cells. The T cell engagers are the most popular BsAbs which account for nearly half of the clinical trials aimed evaluating the BsAbs. NK cell engagers are recently developed BsAbs for NK cell-dependent tumor cell elimination. Bridging receptors is an obligate mechanism in which the binding of BsAbs to 2 receptors causes the activation or inhibition of each receptor. The co-activation or inhibition synergistically enhanced the biological effect of single antibody. In addition to bind to the receptors, targeting redundancy for cytokines or angiogenesis factors represents an area of interest for BsAbs. Instead of targeting two different proteins, bsAbs may be designed to simultaneously bind to two non-overlapping epitopes on the same target. Biparatopic targeting builds on increasing binding strength through antigen crosslinking and aggregation, thereby mimicking effects observed for antibody mixtures and polyclonal antibodies. Biparatopic bsAbs are therefore essentially a combinatorial concept BsAbs can also be designed as a scaffold or cofactor linking enzyme and substrate together. One of the applications is the BsAbs used as a substitution of a critical clotting factor in the treatment of hemophilia. Exploit the first binding specificity of a BsAb as a transport modality for the second specificity are named the “piggyback” approaches. To cross the blood-brain barrier, one binding arm of the BsAbs are designed to target the transferrin receptor (TfR). The human serum albumin (HSA) targeting domain are used to extent the half-life of BsAbs, especially to BsAbs without Fc, for example the tandem ScFvs or VHHs. In addition, the piggyback approaches are also been used in promoting the degradation of pathogens. Bispecific antibodies (BsAbs) are designed to bind two different epitopes or antigens, which leads to multiple mechanistic functions with synergistic effects. The attractive feature is their potential for novel functionalities, which do not exist in mixtures of the parental or reference antibodies. Till now, more than 200 BsAb-based clinical trails have been registered on clinicaltrails.org and 4 BsAbs (with one withdraw) have been granted FDA approval. The promising future makes BsAbs attracting more attentions. Many formats have been developed for BsAb generation as listed in the following table.
1.Good binding of GM-528 to Jurkat cells.
2.High concentration (10ug/ml) of GM-528 should be used for CD3 detection.
3.The CD3 antibody is derived from L2K. The affinity of L2K is about 35 times lower than OKT3. (Therapeutic Window of MuS110, a Single-Chain Antibody Construct Bispecific for Murine EpCAM and Murine CD3. Cancer Res. 2008)
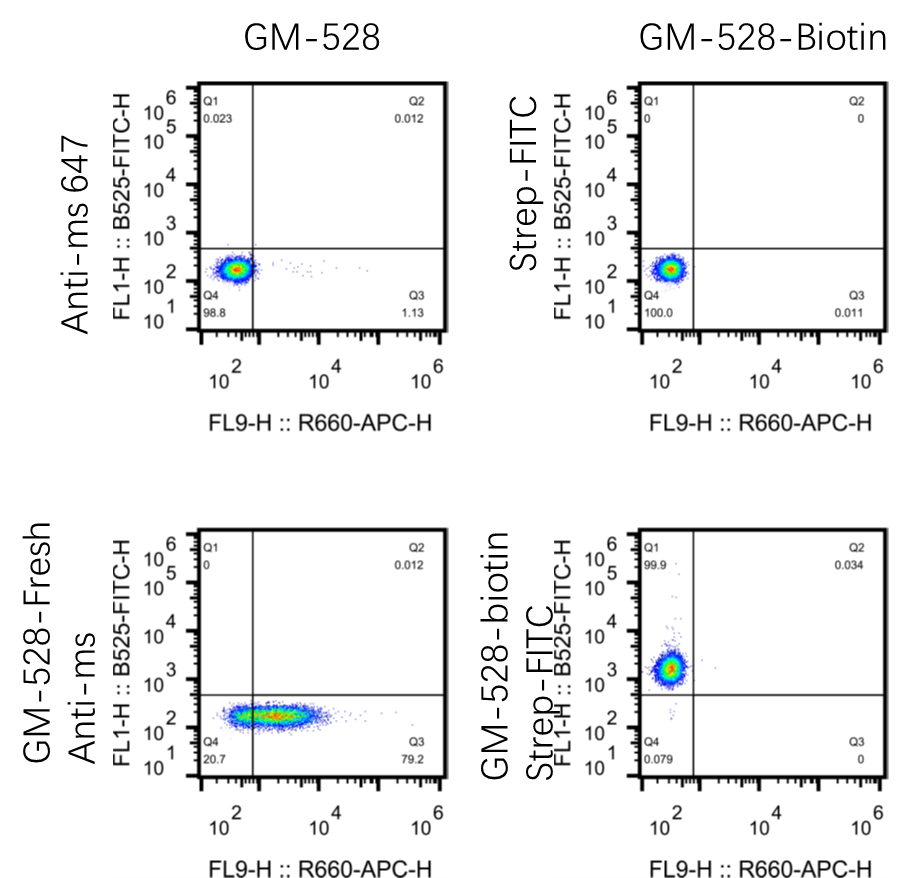
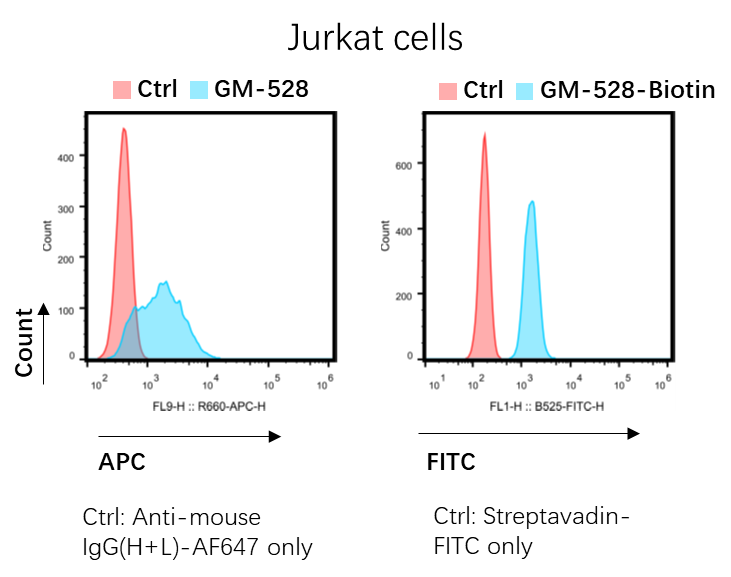
Figure 1. GeneMedi's GMP-Bios-ab-528 (Pre-Made Solitomab biosimilar) is validated to bind the Jurkat cells by Flow Cytometry (FACS Analysis).
Pipeline and MOA (mechanism of action) of bispecific antibodies (BsAbs)
Bridge of 2 cells (engagers)
The engagers in research, clinical trials and market are listed below. Genemedi offers the high quality, premade benchmark BsAbs for researchers.
Targeting multiple receptors
ligand redundancy
Biparatopic bsAbs
Cofactor mimetics
Piggyback approaches
Introduction about bispecific antibodies (BsAbs)
The connecting of two specificities within one BsAb can be exploited for novel therapeutic concepts. The mostly applications of BsAbs are recruiting effector cells to the target cells, which are regarded as the immune cell engagers. The redirection of the cytotoxic effector cells (T and NK cells) to the targeting cancer cells by BsAbs physically link them together and activate the effector cells to eliminate the target ones. In addition, the BsAbs can also be used to link two molecules together to make the therapeutic effects such as the dual inhibition of immune checkpoints, the conditionally activate a growth factor receptor for diabetes treatment and the replacement of natural bridge molecules such as the coagulation factor VIII. More innovative designs of BsAbs can be achieved in the future.
To accelerate the development of BsAbs, Genemedi offers the high quality, premade benchmark BsAbs for researchers. The biosimilars are expressed by mammalian cell line and used for biological drug discovery items including cell culture, assay development, animal model development, PK/PD model development (Pharmacokinetics & Pharmacodynamic) and mechanism of action (MOA) research.
Formats of bispecific antibodies (BsAbs)
Bispecific Antibody Development Programs Guidance for Industry by FDA
FDA set up the guidance for the development of BsAbs. The link and PDF files are listed below.
Bispecific Antibody Development Programs Guidance for Industry






 Products list of Bispecific Antibodies (BsAbs)
Products list of Bispecific Antibodies (BsAbs)