Lipogene Cell Transfection Reagent (both for plasmids transfection & siRNA transfection) FAQs
Order Information
| Package | Catalog No. | Price(In USD) | Qty (Quantity) | Sum(In USD) | 5 ml | R-LG03 | 1125 |
|---|---|---|---|---|
| 10 ml | R-LG04 | 2000 | ||
| 50 ml | R-LG05 | 7500 | ||
| 100 ml | R-LG06 | 12500 | ||
| Sub total: | ||||
| Shipping Cost: | 360.00 | |||
| Total: | ||||
Introduction to LipoGene Transfectin Reagent
LipoGeneTM transfection reagent is highly potent cationic lipofection reagent that has been shown to effectively transfect plasmids or siRNA, as well as nucleic acid-protein complexes, into cultured adherent and suspension cell lines. Researchers use LipoGeneTM Reagent for siRNA- and shRNA-based gene knockdown experiments, as well as for gene expression studies.
Transfection ProtocolProperties
| LipoGene transfection reagent | |
|---|---|
| Quantity/Unit | 1 Vial. |
| Form | Liquid. |
| Sipping and Storage Guidelines | Shipped at 4°C, stored at 4 ° C, effective for 1year (do not freeze). |
| Suitable Cell Type | Established cell lines, hard-to-transfect cells, primary cells, stem cells. |
| Sample Type | Plasmid DNA, RNAi plasmids (shRNA, microRNA), synthetic siRNA. |
| Serum Compatible | Yes. |
Advantages
1. Superior performance for co-transfection of siRNA and plasmid DNA.
2. Proven efficacy in the presence of serum—eliminates the need to change media following transfection.
3. Showed no obvious cytotoxicity for most cells without changing the culture medium for 72 hours.
4. Suitable for both adherent and suspension cells and can be used for screening for stable expression of cell lines.
5. For gene silencing, LipoGeneTM's high-efficiency transfections provide the high levels of gene knockdown needed to achieve convincing results.
6. LipoGeneTM is very easy to use—simply mix with nucleic acid and add to cell culture.
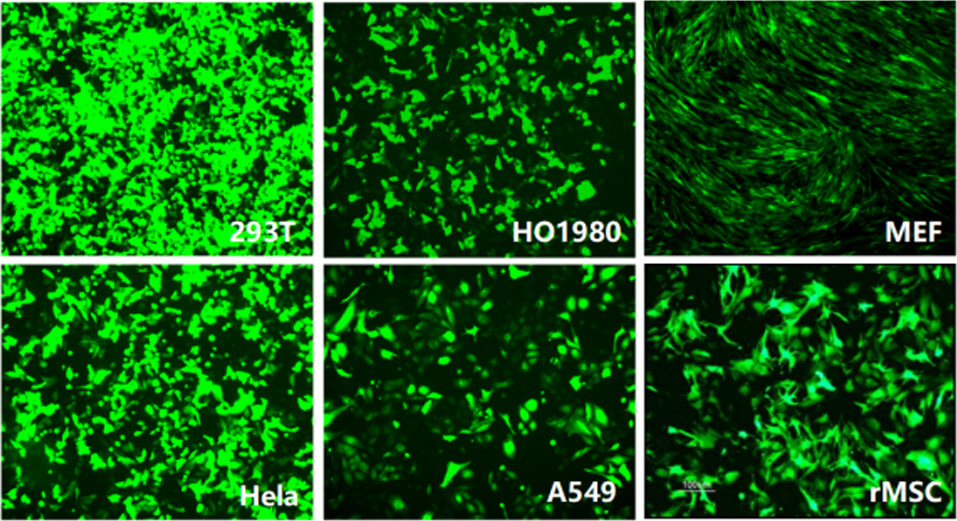
Applications and Figures
Technical Documents
1. For more information please refer to  Lipogene Protocol
Lipogene Protocol
Frequently Asked Questions(FAQs)
- 1. Are cell density (% confluency) and passage number important considerations for transfection?
-
Answer
Yes, cell density is an important parameter in influencing transfection efficiency. If the seeding density is too low, some cytotoxicity may be observed. If the cell density is high, lower than expected transfection efficiency may be observed. Both issues may be easily resolved by either decreasing or increasing the quantity of complexes added to the culture. LipoGene provide excellent transfection efficiencies at confluencies between 70 and 90%. Some toxicity may be observed at lower confluencies but may be alleviated by decreasing quantity of complexes or removing the complexes after 4-6 hours incubation and refreshing the media.
Passage number may affect transfection experiments. We recommend consistent splitting and plating of cells. Excessive numbers of passages may decrease transfection performance. We do not recommend splitting cells for more than 20-30 passages. If transfection performance declines and cells have been in culture for a long time or excessively/improperly passaged, we recommend that you restart your cultures with a new vial of cells from liquid nitrogen.
- 2. Can antibiotics be used in media during transfection?
-
Answer
Yes, antibiotics (penicillin-streptomycin) can be used in the medium during transfection. We have compared transfecting cells in medium with and without antibiotics in multiple cell lines, assessed both the transfection efficiency and toxicity, and found no difference. For certain cell types that are sensitive to transfection or have toxicity issues, omitting antibiotics may improve results, however. For stable transfections, wait at least 48 hours after transfection before adding selective antibiotics.
- 3. Why would the expression level of my gene in transiently transfected cells be greater than those that are stably transfected?
-
Answer
Expression in transiently transfected clones is typically higher because transiently transfected cells can have up to hundreds of copies of the plasmid containing the gene of interest. Stably transfected clones usually harbor 1-2 copies integrated into the genome, and hence have lower levels of expression. Sometimes, the lower expression level in stably transfected cells is due to adverse effects of the recombinant protein on the cell when expressed constitutively.
- 4. Can I use the same amount of any transfection reagent for different cell lines?
-
Answer
The transfection efficiency is highly dependent on the amount of reagent used per well and may be different between reagents. Please consult the product information that is provided with the transfection reagent for optimal use.
- 5. Can I use LipoGeneTM for transfection of suspension cells, other hard-to-transfect cells, and primary cells?
-
Answer
Yes, you can use LipoGeneTM for transfection of suspension cells, hard-to-transfect cells, and primary cells.