Custom Lentivirus Production & Vector System Services
Order Information
-
Step 1:
Enter gene information -
Step 2:
Lentivirus vector construction -
Step 3:
Lentivirus packaging -
Step 4:
Custom Order confirmation
| Catalog No. | Product | Lead time(workday) |
|---|---|---|
| Buy | Lentivirus Vector Production Scale | Final Deliverables | Lead Time(workday) |
|---|
Introduction to Custom Lentivirus Production Service
Lentivirus vector system and lentivirus production service:lentiviral vector(lentivirus plasmids) cloning,
lentivirus packaging, purification,premade lentivirus.
HIV-1 (human immunodeficiency virus type I) based defective lentivirus has been one of the most widely used gene
therapy vectors. It is a powerful tool for introduction of exogenous genes. The most advantageous feature of
lentivirus vectors is to mediate efficient transfection and long-term expression of exogenous genes in both dividing
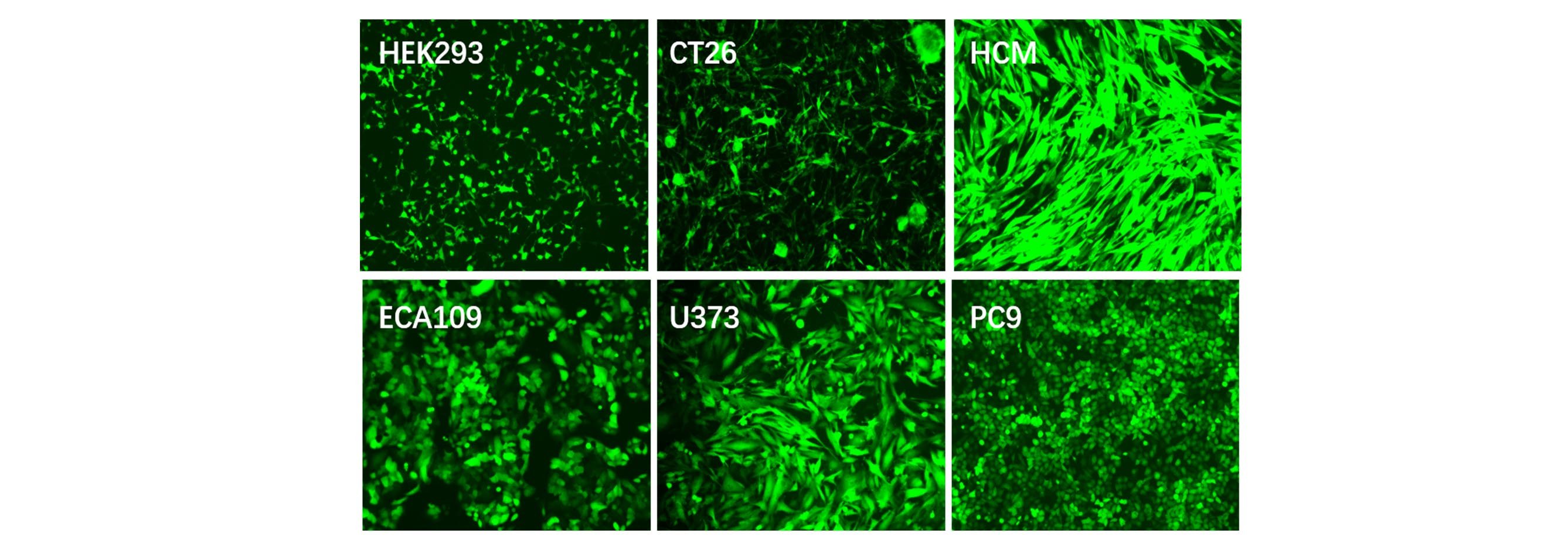
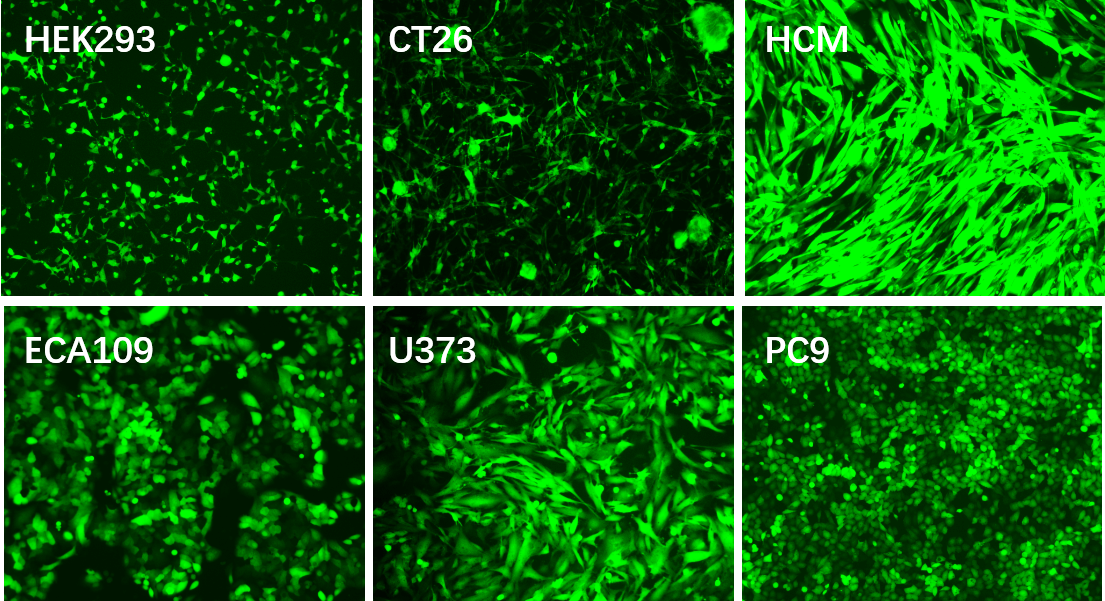
and non-dividing cells. The lentivirus system has been widely used in various cell lines for gene overexpression,
RNA interference, microRNA research and in vivo animal experiments.
Genemed provides lentiviral vector construction, lentivirus packaging for RNA interference, miRNA,
overexpression constructs and stable transfected cell line construction services. Genemed offers high titer, high
purified and ready-to use lentivirus in just two weeks. Please refer to the lentivirus packaging options before you
order.
Flow chart
Properties
| Lentivirus custom packaging | |
|---|---|
| Quantity/Unit | 1 Vials. |
| Form | Frozen form. |
| Sipping and Storage Guidelines | Shipped by dry ice, stored at -80 ° C, effective for 1 year. Avoid repeatedly freezing and thawing. |
| Titer | > 1*10^8 TU/ml. |
Advantages
1. Customized cloning for any other gene ORF expression, shRNA/miRNA and CRISPR/Cas9.
2. Ready-to-use, high purified lentivirus.
3. High titer: 108TU/ml or 109TU/ml lentiviral titer for cell line transfection in medium or large scale.
4. A wide range of host: mediate efficient transfection in both dividing and non-dividing cells.
5. Stable expression: mediate long-term expression of exogenous genes.
Applications and Figures
Quality control description
Our optimized production of lentiviral vector and strict quality control systems supply customers with a high titer of functional recombinant lentiviral vectors. Two methods are employed to determine viral titers: physical titer (VP/mL) and functional titer (TU/mL). Physical titer is calculated by the level of protein, such as p24, or viral nucleic acid. The functional titer, a calculation of the active virus that can infect cells, is much less than the physical titer (100-1000 fold lower). The method we adopted is functional titer, which is an accurate solution for testing virus accurate activity and MOI. The physical titer can only reflect the number of virus particles, but not reflect the true viral activity, which will cause a large error in subsequent infection experiments.
Technical Documents
1. For further information about lentivirus administration and transduction, please see  Lentivirus User Manual.
Lentivirus User Manual.
Frequently Asked Questions(FAQs)
- 1. How are Genemedi lentiviral vectors different from other viral vectors?
-
Answer
Genemedi has a particular know-how in the field of lentiviral vector design and production stemming from years of research. This experience has led us to set up optimized protocols and reach high quality standards, constantly improved. Hence, Genemedi lentiviral vector productions show high efficiency, suited for either in vivo, in vitro or transgenesis applications. Our expertise allows us to take on all challenges. Notably, we provide our clients with lentiviral vectors expressing particularly challenging transgenes, for instance, ones that directly interfere with the lentiviral cycle.
- 2. For what kind of applications can lentiviral vectors be used?
-
Answer
Our lentiviral vectors can be used in large array of purposes. Taking advantage of our products can significantly enhance your research. Here are some examples of in vitro and in vivo applications:
1. Stable expression transgenes of interest in target cells.
2. Expression of reporter systems to characterize a given vector in particular contexts.
3. Transient expression of transgenes of interest in dividing cells.
4. Induction of long term expression in non-dividing cells with reduced genotoxicity.
5. Generation of cellular models, such as cell lines with recombination induced by transiently expressed genome editing tools (eg: CRISPR).
6. Generation of animal models by zygote injection and transient expression of genome editing tools (KO animals). - 3. Which cell type is transduced by Genemedi?
-
Answer
Lentiviral vectors are very flexible gene transfer tools and can transduce a large variety of cells. Genemedi can mediate ubiquitous or cell specific expression depending on the envelope and the promoter selected for your vector. Indeed the envelope will determine the entry mechanism of the vector into the cell, and the promoter will define the mechanism of transcription of the transgene. Consequently, the combination of these elements generates ubiquitous transduction and transgene expression in a wide range of cell types.
- 4. What are the advantages of lentivirus over retrovirus and adenovirus?
-
Answer
Lentiviruses comprise a subtype of retroviruses. Lentiviruses can stably integrate into the host genome in dividing, non-dividing and post-mitotic mammalian cells, while retroviruses are less active in this scenario. Adenoviruses can also transduce non-dividing cells, but can’t stably integrate into the host cell’s genome. Adenoviruses also take much more time to design and prepare. In addition, lentiviruses are much less immunogenic than the retroviruses and adenoviruses, making lentivirus more suitable for use in most types of cells and animal models.
- 5. What are the storage and handling recommendations?
-
Answer
Upon receipt, the viral vectors must be stored at -80°C.
The vectors should be taken out from the -80°C freezer at the last moment and kept on ice for gentle thawing. Once thawed, they should be used for transduction as soon as possible to avoid degradation. It is essential to avoid heat shock of the viral vectors and cells. If the vectors should be diluted in medium, use a medium at room temperature to minimize the heat shock experienced by both vectors and cells.
We recommend not refreezing the viral vectors. In order to guarantee viral vectors titer, we recommend keeping freeze-thaw cycles to a minimum. In case that more than one freeze-thaw cycle is required according to your application, Genemedi recommends applying a decrease of about 15% on the titer for each freeze-thaw cycle.
- 6. What are the safety issues when using Genemedi lentiviral vectors?
-
Answer
Genemedi vectors are biosafety level 2 products. The use of retroviral derived vectors implies laboratory biosafety procedures and practices in accordance with regulations applicable in your country.
Genemedi vectors are suited for research applications, either in vitro or in vivo, providing that you respect local laws regarding GMO class 2 storage and handling. All Genemedi vectors are distributed for laboratory research use only and cannot under any circumstances be used for diagnostic or treatment applications. For more information on biosafety levels please visit http://www.cdc.gov/od/ohs/biosfty/bmbl5/bmbl5toc.htm.
- 7. What is virus titer? How is lentiviral stock titer determined?
-
Answer
In strict sense, the definition of lentiviral vector titer is the number of lentivirus particles required to infect a cell, present in a volume. Titration of lentiviral preparation is important to consistent and reliable experiment result. Two classes of titration methods have been described for evaluation of lentiviral vector titers, including functional and physical titration methods. Functional assay includes determination of transducing units following transducing cells with limiting dilution of lentiviral preparation, assessment of the number of colony forming units following antibiotic selection, as well as FACS when fluorescent reporters are expressed. The latter includes determination of the quantity of p24 antigen by ELISA, reverse transcriptase activity and the genomic RNA concentration in vector preparation. The physical methods provide the information of the number of physical lentivirus particles in lentiviral preparation. Functional assay generates valuable data on the quality of these lentiviral vector particles.
- 8. What's the optimal concentration of viruses that I should use for infection?
-
Answer
It depends on the purpose of the experiment. Higher titer of lentivirus should be used for in vivo experiment compared to in vitro experiment. Though lentiviral vector is highly efficient in transduction, the transduction efficiency of lentivirus depends heavily on the cell type to be transduced. Pilot experiment is highly recommended to determine how efficient the lentivirus on your target cells.
- 9. How much culture media should I use during infection?
-
Answer
For your reference, we recommend the following amount of virus-containing media for infection:
1. 10-cm plate: 8-10 ml/plate.
2. 6-well plate: 1 ml/well.
3. 12-well plate: 0.5 ml/well.
4. 24-well plate: 0.2 ml/well. - 10. How do I determine the correct amount of lentivirus to add to the target cells?
-
Answer
One of the key factors of a successful transduction is the cell type. For example, transduction efficiency is much higher in actively dividing cells than in non-dividing cells. In addition, transduction of cells works better at lower MOI (multiplicity of infection) than at higher MOI. MOI is the ratio of the number of lentivirus particles to the number of cells. For some cell types, the higher the MOI , the larger the volume and higher the titer of lentivirus is required in order for the experiment to succeed. You can adjust the cell number and add the appropriate amount of lentivirus according to what has been reported in the scientific literature. If there is no adequate information in the scientific literature, we recommend performing a preliminary experiment using gradient dilutions of lentivirus, such as 0.1 μl, 0.3 μl, 0.5 μl, 0.7 μl, 0.9 μl for Genemedi purified particles. Another important consideration for getting good transduction efficiency is the cell status. Transduction efficiency varies greatly between healthy cells and unhealthy cells. Therefore, it is essential to keep the cells as healthy as possible. For some cells with high MOI, you could also include additives such as polybrene to enhance the transduction efficiency. However, the overall health of the cells itself is always the most essential element.
- 11. Does freeze-thaw cycle infect the titer of lentiviruses?
-
Answer
Yes, multiple freeze-thaw cycles may reduce the functional titer of the virus stock by up to 2-4 folds. However, first one freeze-thaw cycle does not lower the lentiviral titer. Genemedi recommends our customer not to use lentivirus that has gone through more than two freeze-thaw cycles for titer reason.
- 12. Why do my cells die in quantity after adding lentivirus to the culture medium?
-
Answer
Lentivirus has some level of toxicity to cells. It may cause damage to your cell of interest with either superfluous amounts of lentivirus, or if the infection were allowed to go on for too long a period of time. In these cases, you can adjust the multiplicity of infection (MOI) to a lower range. We recommend replacing the old culture medium with fresh complete medium 4-8 hours post transduction (no later than 12 hours post transduction).
- 13. What is the capacity of lentiviral vector as an expression system?
-
Answer
The cloning capacity for the transgene is approximately 3-4 kb for most vector formats.
- 14. How do I create a stable cell line using lentivirus?
-
Answer
Lentiviruses can stably integrate into the host cell’s genome and obtain a consistent level of expression. With a selectable marker in the lentiviral gene transfer vector plasmid, it is easy to generate a stable cell line using drug selection. You can use qRT-PCR, western blot or other detection methods to estimate the expression level of your gene.
- 15. How custom lentivirus production service proceeds
-
Answer
If lentiviral construct is ready, please provide us:
1. The construct map or sequence.
2. 1~5 µg of your lentiviral construct plasmid DNA or bacterial culture.
We will prepare Endotoxin-free plasmid and transfect our packaging cell line for your lentivirus production.
Lentiviral particles will be harvested followed by concentration and titration.
For 200 ul of lentivirus production, you will receive eight 25 µl aliquots.If you need us to generate lentiviral transfer vector for your target gene, please:
1. Choose lentiviral vector format from our vector collection and specify the gene of interest.
2. Please send us 1~5 µg plasmid or bacterial culture for your gene if it is available, otherwise please send us cDNA sequence.
We will subclone the gene of interest or shRNA into the chosen lentiviral vector and confirm its accuracy by sequencing. - 16.How long does the service take? Is my project too big (or too small)?
-
Answer
Get ready-to-transduce, high-quality, high titer lentiviral preparations at the production scale your project needs—even at large scales of up to 10 mL. Use your own lentivector construct or take advantage of our Custom Construct services and we’ll handle vector construction as well. We even have an ultra-high titer offering for demanding applications such as in vivo and stem cell transductions.
Reference
1. Wu, J. et al. MicroRNA-30 family members regulate calcium/calcineurin signaling in podocytes. Journal of Clinical Investigation 125, 4091-4106 (2015).
2. Li, F., Li, S. & Cheng, T. TGF-β1 Promotes Osteosarcoma Cell Migration and Invasion Through the miR- 143-Versican Pathway. Cellular Physiology and Biochemistry 34, 2169-2179 (2014).
3. Liu, Z. et al. miR-451a Inhibited Cell Proliferation and Enhanced Tamoxifen Sensitive in Breast Cancer via Macrophage Migration Inhibitory Factor. BioMed Research International 2015, 207684-207684 (2015).
4. Si, L. et al. Smad4 mediated BMP2 signal is essential for the regulation of GATA4 and Nkx2.5 by affecting the histone H3 acetylation in H9c2 cells. Biochemical and Biophysical Research Communications 450, 81-86 (2014).
5. Han, H., Yang, S., Lin, S. G., Xu, C. S. & Han, Z. Effects and mechanism of downregulation of COX‑2 expression by RNA interference on proliferation and apoptosis of human breast cancer MCF‑7 cells. Molecular Medicine Reports 10, 3092-3098 (2014).
6. Zhang, G., Liu, Z., Cui, G., Wang, X. & Yang, Z. MicroRNA-486-5p targeting PIM-1 suppresses cell proliferation in breast cancer cells. Tumor Biology 35, 11137-11145 (2014).
7. Li, G. et al. CYC1 silencing sensitizes osteosarcoma cells to TRAIL-induced apoptosis. Cellular Physiology and Biochemistry 34, 2070-2080 (2014).
8. Mao, J., Lv, Z. & Zhuang, Y. MicroRNA-23a is involved in tumor necrosis factor-α induced apoptosis in mesenchymal stem cells and myocardial infarction. Experimental and Molecular Pathology 97, 23-30 (2014).
9. Liu, X. et al. Role of human pulmonary fibroblast-derived MCP-1 in cell activation and migration in experimental silicosis. Toxicology and Applied Pharmacology 288, 152-160 (2015).
10. Guan, G. et al. CXCR4-targeted near-infrared imaging allows detection of orthotopic and metastatic human osteosarcoma in a mouse model. Scientific Reports 5, 15244-15244 (2015).
11. Zhang, Y. et al. Role of high-mobility group box 1 in methamphetamine-induced activation and migration of astrocytes. Journal of Neuroinflammation 12, 156-156 (2015).
12. Zhu, T. et al. The Role of MCPIP1 in Ischemia/Reperfusion Injury-Induced HUVEC Migration and Apoptosis. Cellular Physiology and Biochemistry 37, 577-591 (2015).
13. Qian, M. et al. P50-associated COX-2 extragenic RNA (PACER) overexpression promotes proliferation and metastasis of osteosarcoma cells by activating COX-2 gene. Tumor Biology 37, 3879-3886 (2016).
14. Wu, N., Song, Y., Pang, L. & Chen, Z. CRCT1 regulated by microRNA-520 g inhibits proliferation and induces apoptosis in esophageal squamous cell cancer. Tumor Biology 37, 8271-8279 (2016).
15. Wang, Y. et al. Overexpression of Hiwi Inhibits the Growth and Migration of Chronic Myeloid Leukemia Cells. Cell Biochemistry and Biophysics 73, 117-124 (2015).
16. Niu, L. et al. RNF43 Inhibits Cancer Cell Proliferation and Could be a Potential Prognostic Factor for Human Gastric Carcinoma. Cellular Physiology and Biochemistry 36, 1835-1846 (2015).
17. Zhang, H. et al. ZC3H12D attenuated inflammation responses by reducing mRNA stability of proinflammatory genes. Molecular Immunology 67, 206-212 (2015).
18. Deng, X. et al. MiR-146b-5p Promotes Metastasis and Induces Epithelial-Mesenchymal Transition in Thyroid Cancer by Targeting ZNRF3. Cellular Physiology and Biochemistry 35, 71-82 (2015).
19. Zhang, B. et al. HSF1 Relieves Amyloid-β-Induced Cardiomyocytes Apoptosis. Cell Biochemistry and Biophysics 72, 579-587 (2015).
20. Hu, Q. et al. Periostin Mediates TGF-β-Induced Epithelial Mesenchymal Transition in Prostate Cancer Cells. Cellular Physiology and Biochemistry 36, 799-809 (2015).
21. Yang, Z. et al. CD49f Acts as an Inflammation Sensor to Regulate Differentiation, Adhesion, and Migration of Human Mesenchymal Stem Cells. Stem Cells 33, 2798-2810 (2015).
22. Wang, X. et al. MCPIP1 Regulates Alveolar Macrophage Apoptosis and Pulmonary Fibroblast Activation After in vitro Exposure to Silica. Toxicological Sciences 151, 126-138 (2016).
23. Gu, S., Ran, S., Liu, B. & Liang, J. miR-152 induces human dental pulp stem cell senescence by inhibiting SIRT7 expression. FEBS Letters 590, 1123-1131 (2016).
24. Jin, F., Qiao, C., Luan, N. & Li, H. Lentivirus-mediated PHLDA2 overexpression inhibits trophoblast proliferation, migration and invasion, and induces apoptosis. International Journal of Molecular Medicine 37, 949-957 (2016).
25. Liu, Z., Song, Y., Wan, L., Zhang, Y. & Zhou, L. Over-expression of miR-451a can enhance the sensitivity of breast cancer cells to tamoxifen by regulating 14-3-3ζ, estrogen receptor α, and autophagy. Life Sciences 149, 104-113 (2016).
26. Tian, Y. et al. MicroRNA-30a promotes chondrogenic differentiation of mesenchymal stem cells through inhibiting Delta-like 4 expression. Life Sciences 148, 220-228 (2016).
27. Xu, S. et al. MicroRNA-33 promotes the replicative senescence of mouse embryonic fibroblasts by suppressing CDK6. Biochemical and Biophysical Research Communications 473, 1064-1070 (2016).
28. Chen, H., Sun, M., Liu, J., Tong, C. & Meng, T. Silencing of Paternally Expressed Gene 10 Inhibits Trophoblast Proliferation and Invasion. PLOS ONE 10 (2015).
29. Deng, Y. et al. Repair of critical-sized bone defects with anti-miR-31-expressing bone marrow stromal stem cells and poly(glycerol sebacate) scaffolds. European Cells & Materials 27, 13-25 (2014).
30. Zheng, Y. & Xu, Z. MicroRNA-22 induces endothelial progenitor cell senescence by targeting AKT3. Cellular Physiology and Biochemistry 34, 1547-1555 (2014).
31. Yang, X. et al. A lentiviral sponge for miRNA-21 diminishes aerobic glycolysis in bladder cancer T24 cells via the PTEN/PI3K/AKT/mTOR axis. Tumor Biology 36, 383-391 (2015).
32. Wang, W. et al. p53/PUMA expression in human pulmonary fibroblasts mediates cell activation and migration in silicosis. Scientific Reports 5, 16900-16900 (2015).
33. Zhang, S. & Qi, Q. MTSS1 suppresses cell migration and invasion by targeting CTTN in glioblastoma. Journal of Neuro-oncology 121, 425-431 (2015).
34. Wang, P. et al. PFDN1, an indicator for colorectal cancer prognosis, enhances tumor cell proliferation and motility through cytoskeletal reorganization. Medical Oncology 32, 264-264 (2015).
35. Gu, S. et al. Human Dental Pulp Stem Cells via the NF-κB Pathway. Cellular Physiology and Biochemistry 36, 1725-1734 (2015).
36. Huang, G. et al. Clinical and therapeutic significance of sirtuin-4 expression in colorectal cancer. Oncology Reports 35, 2801-2810 (2016).
37. Yan, X., Ye, T., Hu, X., Zhao, P. & Wang, X. 58-F, a flavanone from Ophiopogon japonicus, prevents hepatocyte death by decreasing lysosomal membrane permeability. Scientific Reports 6, 27875 (2016).
38. Ding, W., Tong, Y., Zhang, X., Pan, M. & Chen, S. Study of Arsenic Sulfide in Solid Tumor Cells Reveals Regulation of Nuclear Factors of Activated T-cells by PML and p53. Scientific Reports 6, 19793-19793 (2016).