Tandem VHH/Nanobody (single domain antibody): Structure, Production, Application and Products
Since 1975, monoclonal antibodies have been successfully used for the diagnosis and therapeutics. In 1993, heavy-chain-only antibodies which bind to antigens was discovered in camelids. The variable domain of heavy chain antibody (VHH) was called as nanobodies and it was anticipated as the better therapeutic antibodies in future due to its high stability, refoldability, and manipulable characteristics. VHH has also been effectively used for the diagnosis, bioimaging, immunoassay and affinity purification.
GeneMedi's products list of Nanobodies (single domain antibodies)
| Cat No. | Products Name (INN Index) | INN Name | SAbDab Target/Gene Name | Format |
| GMP-Bios-ab-093 | Pre-Made Caplacizumab biosimilar, Nanobody, Anti-VWF Antibody: Anti-F8VWF/VWD therapeutic antibody | Caplacizumab | VWF | Nanobody |
| GMP-Bios-ab-188 | Pre-Made Envafolimab biosimilar, Single Domain Variable Fragment;H, Anti-CD274/PD-L1 Antibody: Anti-B7-H, B7H1, PD-L1, PDCD1L1, PDCD1LG1, PDL1, hPD-L1 therapeutic antibody | Envafolimab | PDL1 | Single Domain Variable Fragment;H |
| GMP-Bios-ab-307 | Pre-Made Letolizumab biosimilar, Single Domain Variable Fragment;H, Anti-CD40LG Antibody: Anti-IGM/IMD3/TRAP/gp39/CD154/CD40L/HIGM1/T-BAM/TNFSF5/hCD40L therapeutic antibody | Letolizumab | CD40LG | Single Domain Variable Fragment;H |
| GMP-Bios-ab-326 | Pre-Made Lulizumab biosimilar, Single Domain Variable Fragment;L, Anti-CD28 Antibody: Anti-Tp44 therapeutic antibody | Lulizumab | CD28 | Single Domain Variable Fragment;L |
| GMP-Bios-ab-445 | Pre-Made Placulumab biosimilar, Single Domain Variable Fragment (VL + Fc), Anti-TNFA/TNF Antibody: Anti-DIF/TNFSF2/TNLG1F therapeutic antibody | Placulumab | TNFA | Single Domain Variable Fragment (VL) + Fc |
| GMP-Bios-ab-689 | Pre-Made Rimteravimab biosimilar, Whole Mab: Anti-SARS-CoV-2 Spike RBD therapeutic antibody | Rimteravimab | SARS-CoV-2 Spike RBD | Mixed Nanobody (VHH-CH2-CH3 dimer) |
Structure and Physicochemical Features of Nanobody
Nanobody (single domain antibody) (Nb) is the smallest natural antigen-specific binding functional fragment of about 12~15 kb (Figure 2D). The molecular weight of Nb is smaller than the monoclonal antibody mAb (~150 kb), Fab fragment (~55 kb) or HcAb (~90 kb) Figure 1A–C. Former study showed that these nanobodies can be genetically engineered from the heavy-chain antibody derived from camelids or cartilaginous fish. Their immune systems have naturally evolved into high-affinity V-like domains. Nb has a stronger and faster tissue penetration ability and can reach dense tissues such as solid tumors. The half-life of Nb in the blood is also relatively shorter due to renal filtration.
Nb consists of three antigenic complementary determining regions (complementarity determining region, CDR) and four frame regions (frame region, FR). Among them, three CDR are the binding regions of Nb to the antigen. The longer amino acid sequences of CDR1 and CDR3 of Nb make up for the loss of antigen-binding ability caused by the loss of light chain to some extent. After receiving antigen stimulation, the production of Nb mainly depends on somatic hypermutation, so a longer CDR sequence also means more antibody diversity. Crystallographic studies have shown that longer CDR3 regions give Nb a stronger ability to bind antigens. Sequence analysis disclosed that the VH domain of Nb was highly homologous to that of human immunoglobulin IgG, but there were significant differences between FR2 and CDR3 regions. Repeated administration of Nb could induce both humoral and cellular immune response.
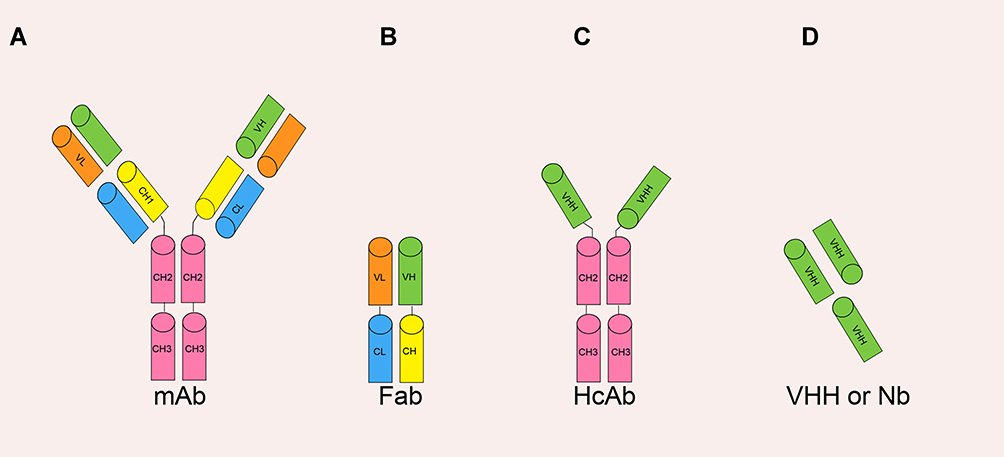
Figure 2 The various antibody formats: (A) mAb (monoclonal antibody); (B) Fab (fragment antigen binding); (C) HcAb (camel heavy-chain antibody); (D) VHH or Nb (nanobody) (adopted from Yang et al. 2019).
Nanobody (single domain antibody) production
The production of nanobody primarily required to select a camelid animal for immunization for about 5–10 weeks. The effector B cells are extracted from the plasma peripheral lymph blood of the immunized camel to isolate the total RNA and cDNA synthesis for sequencing. Phage display technology is used to screen the specific binding ability of the nanobody. Finally, the phage vector was transferred to the Nb expression system to express the Nb proteins. Even though the Nb production technology is relatively complicated, the application of phage display technology and the screening steps of Nb are being gradually optimized.
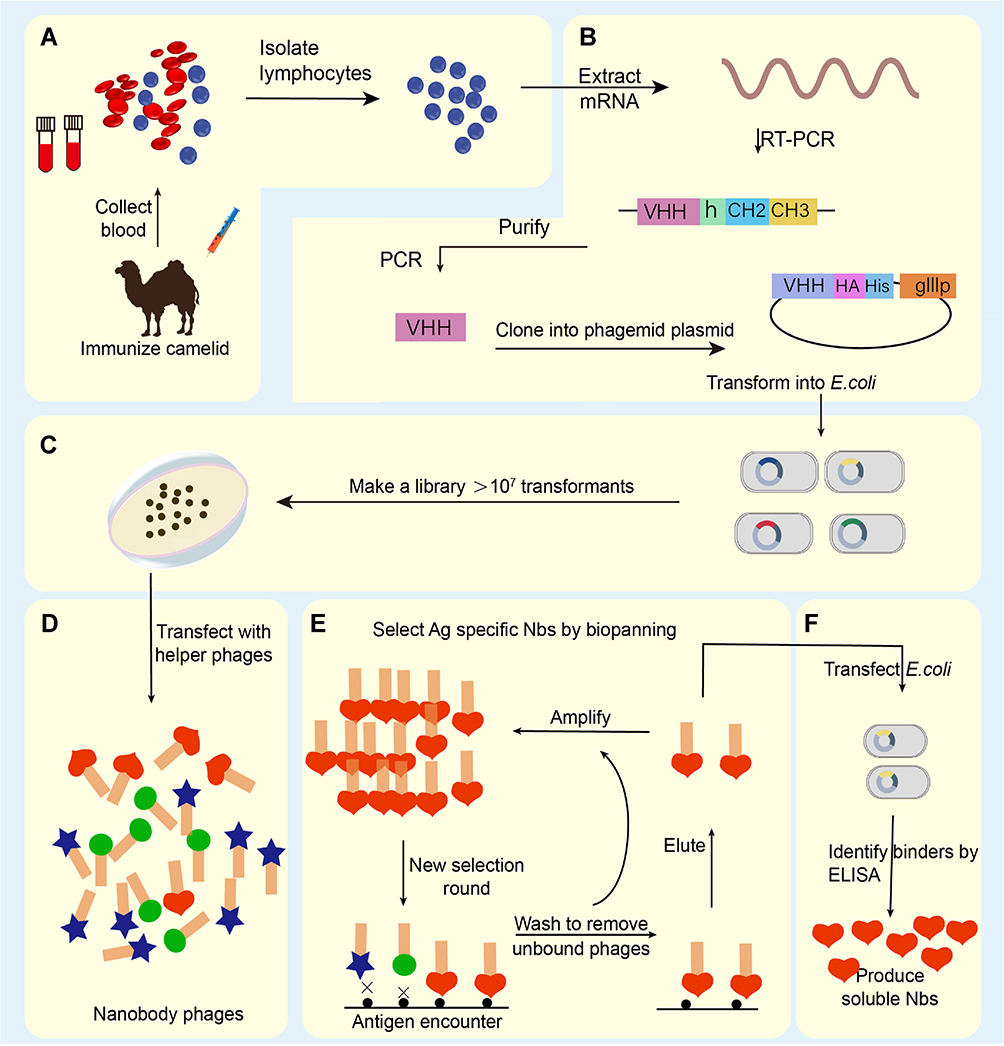
Figure 1 Schematic overview of nanobody generation. (A) Extracting a cDNA from peripheral blood obtained from a camelid after immunized. (B) Extracting Total mRNA and reversely transcribing into cDNA, then amplifying the VHH, the phage display vector and the VHH are digested by restriction enzymes, and the two fragments are connected. (C) Constructing natural or immune camelid-derived nanobody libraries. (D–F) Obtaining antigen-specific nanobody by multiple screening (adopted from Sun et al. 2021).
Application of nanobodies
Nanobodies have been used for both diagnostic and therapeutic application due to its high stability, refoldability, and manipulable characteristics. In addition, VHH has been effectively used as a research tool.
1. In Vitro Diagnostic Tests
Nbs have been recommended for both in vitro as well as in vivo diagnostic tests. Early diagnosis of both infection and disease are important for the disease recovery. The various Nb-based in vitro diagnostic assay have been developed to identify the cancers and viral infectious diseases at early stages. Nbs have been used to screen possible contaminations such as toxins, pesticides, and herbicides in food or other consumer goods. Thus, Nbs have been used in ELISA to detect biomarker proteins, viruses, or other pathogens, and organic pesticides. Diagnostic sensitivity has been increased by capturing Nb bivalent or trivalent and by biotin tagging at its C-terminal end for directional immobilization on coated streptavidin and to improve the exposure of the antigen-capturing Nb into the solution. Further, Nbs have been expressed as fusions with alkaline phosphatase, horseradish peroxidase, or nanoluciferase to increase the sensitive. Competitive inhibition ELISA is used to detect the pesticides or toxin content in the food. In addition, Nb based lateral flow assay (LFA) is much more convenient for preliminary screening.
2. In Vivo Diagnostic Tests
Nbs are used as a tracer for noninvasive imaging. Nbs allow an efficient labeling with short-lived γ- or positron-emitting nuclides. Due to the small size, intravenously injected Nbs in tumor-bearing mice will rapidly extravasate and diffuse in tissues to trace the cognate antigens. Therefore, SPECT (single-photon emission computerized tomography) or PET/CT (positron emission tomography/computerized tomography) imaging can be obtained very soon after tracer administration. Nb directed against breast carcinoma HER2 antigen labeled with 68Ga was used to detect the primary breast carcinoma lesions or metastatic lesions. 99mTc-labeled Nb against vascular cell adhesion molecule-1 (VCAM-1) has been used as a biomarker for vulnerable atherosclerotic plaques. various Nbs were developed to monitor arthritis in mouse model.
3. Therapeutic application
The Nbs have broad therapeutic application. Nbs against amyloidogenic proteins prevent the formation of toxic protofibrils. Nbs against A-β potentially neutralize their toxic effect on neuronal cell lines. Nbs against β-microglobulin is probably more realistic. Multiple Nbs have been generated to neutralize various venoms and toxins. Viral infections have been treated more effectively with monoclonal antibodies (mAbs). To combat viral or bacterial infections in animals and humans, Nbs have been reconstituted with the human hinge and Fc region of IgG isotypes of the infected species to restore a HCAb. Instead, the pathogen-specific Nb can be conjugated to a second Nb that targets IgG or (human) serum albumin for passive vaccination. HCAb assemblies with multiple Nbs targeting various viral serotypes or multiple epitopes achieve the protection against virus. Nbs conjugated with Pseudomonas exotoxin A have been used to kill VEGFR2-expressing cancer cells. An antibody-dependent enzyme prodrug therapy (ADEPT) using anti–carcinoembryonic antigen Nbs seemed to be effective at eliminating solid cancers. Nb therapy could reduce toxicity in the healthy tissues.
4. Nanobodies (single domain antibodies) As Research Tools
Nbs are easily captured on beads via their His tag or in vivo biotinylating on commercial streptavidin-coated surfaces and used to purify the recombinant antigenic protein with high specificity. Nbs are also used for the structural characterization of their cognate antigen. Proteins are sometimes difficult to crystallize. Hence, the Interaction between the Nb and its cognate antigen facilitates the crystallization for X-ray diffraction. Nbs are also used to study the protein dynamics using nuclear magnetic resonance. The genes encoding a Nb with a fluorescent protein are used to track the cognate antigen of the Nb within living cells. Fluorescently labeled Nbs have also been used in confocal microscopy. The presence of a membrane-attachment tag immobilizes the Nb on the cell surface and thus preventing antigen spread from this cell. Nb localization could be used to develop a real-time protein–protein interaction monitoring system inside the cellular compartment. Recently, Nbs have been engineered into photobodies or optobodies.
Advantages and limitation of Nanobodies (single domain antibodies)
Nanobodies are frequently compared to polyclonal, monoclonal antibodies, antibody fragments, and small molecules. The production of nanobodies is simple and cost effective compared to the polyclonal antibodies. Besides, Nbs are stable in a wide range of temperatures and pH levels.
Nanobodies are also compatible with genetic engineering methods, which allow scaffolding and alteration of amino acids to improve binding. Relating to structure, the hydrophilic side of nanobodies, that is not present in conventional antibodies, means they do not have issues with solubility and aggregation. The conventional antibodies do not bind well in grooves or cavities on the surface of the antigen. But, Nanobodies bind with the clefts of the active site of antigen.
Since, nanobodies still have some limitations and disadvantages. The nanobodies can be developed only from camelids and sharks. Traditional monoclonal antibodies, on the other hand, are obtained from mice. Therefore, the development of nanobodies requires larger, more complicated housing and animal husbandry for obtaining the desired antibody. The advantages and limitations of nanobodies and antibodies are shown in table 1.
| Antibodies | Nanobodies | |
| Size | 150 kD | 15 kD |
| Development costs | High | Moderate |
| Administration | i.v., s.c. | i.v., s.c., aerosol, topical |
| Specificity | High | High |
| Off target adverse effects | None | None |
| In Vivo half-life | Can be adjusted by Fc-engineering | Can be adjusted by PEGylation or fusion to albumin-specific Nb |
| Metabolites | Non-toxic, biodegradable | Non-toxic, biodegradable |
| Tissue penetration | Slow | Excellent in periphery |
| Tissue specificity | Targetable (bi-specific Abs) | Targetable (bi- specific Nbs) |
| Albumin binding | Usually not | Via albumin-specific Nb to extend half-life, usually no effect on potency |
References
Els B, Jan G, (2017) Nanobody Technology: A Versatile toolkit for microscopic imaging, protein–protein interaction analysis, and protein function exploration. Frontiers in Immunology, 8:771.
Bannas P, Julia H, Friedrich K.N. (2017) Nanobodies and nanobody-based human heavy chain antibodies as antitumor therapeutics. Frontiers in Immunology. 8:1603.
Muyldermans S. (2021) Applications of Nanobodies. Annu Rev Anim Biosci. 16;9:401-421.
Sun S, Ding Z, Yang X, Zhao X, Zhao M, Gao L, Chen Q, Xie S, Liu A, Yin S, Xu Z, Lu X. (2021) Nanobody: A small antibody with big implications for tumor therapeutic strategy. Int J Nanomedicine. 16:2337-2356.
Stephan M, Nicole S, Friedrich H, Friedrich K.N. (2018). Nanobody-based biologics for modulating purinergic signaling in inflammation and immunity. Frontiers in Pharmacology. 09: 266.
Yang X, Xie S, Yang X, et al. (2019) Opportunities and challenges for antibodies against intracellular antigens. Theranostics. 9(25):7792–7806.






