Fetal fibronectin (fFN) antibody and antigen (Recombinant protein) GMP-h-fFN
Diagnostic Fetal fibronectin (fFN) antibodies (anti-Fetal fibronectin, anti-fFN) and antigens (recombinant Fetal fibronectin (fFN) protein) for Fertility fFN detection in ELISA, colloidal gold-based Lateral flow immunoassay (LFIA), CLIA, TINIA and POCT.
Product information
Size: 1mg | 10mg | 100mg
| Catalog No. | Description | US $ Price (per mg) | First Order Discount | First Order Discount Price |
|---|---|---|---|---|
| GMP-h-fFN-Ag01 | Recombinant human FN1/fFN/FN Protein | 3090 | ||
| GMP-h-fFN-Ab01 | Anti-human FN1/fFN/FN mouse monoclonal antibody (mAb) | 1953 | 20% | 1562.4 |
| GMP-h-fFN-Ab02 | Anti-human FN1/fFN/FN mouse monoclonal antibody (mAb) | 1953 | 20% | 1562.4 |
| GMP-h-fFN-Ab03 | Anti-human FN1/fFN/FN mouse monoclonal antibody (mAb) | 1953 | 20% | 1562.4 |
| GMP-h-fFN-Ab04 | Anti-human FN1/fFN/FN mouse monoclonal antibody (mAb) | 1953 | 20% | 1562.4 |
| GMP-h-fFN-Ab05 | Anti-human FN1/fFN/FN mouse monoclonal antibody (mAb) | 1953 | 20% | 1562.4 |
Shipping Costs: $360–$760
Antibodies: $360 Antigens: $760 (Elevated cost due to antigen heterogeneity, post-translational modifications, structural complexity, and specialized handling.)
Product Description
| Cat No. of Products | GMP-h-fFN-Ag01 |
| Product Name | Recombinant human FN1/fFN/FN Protein |
| Target/Biomarker | Fetal fibronectin (fFN) |
| Alias of Target/Biomarker | CIG, ED-B, FINC, FN, FNZ, GFND, GFND2, LETS, MSF, SMDCF |
| Expression platform | mammalian |
| Bioactivity validation | Fetal fibronectin (fFN) antibodies binding, Immunogen in Sandwich Elisa, lateral-flow tests, and other immunoassays as control material in fFN level test of Fertility (premature delivery) and related syndrome evaluation. |
| Tag | His |
| Products description | Recombinant human FN1/fFN/FN Protein was expressed in mammalian expression system and is expressed with 6 HIS tag at the C-terminus. |
| Purity | Purity: ≥95% (SDS-PAGE) |
| Application | positive control, ELISA test,Lateral flow immunoassay (LFIA),colloidal gold immunochromatographic assay, Chemiluminescent immunoassay (CLIA),turbidimetric inhibition immuno assay (TINIA) and Immunonephelometry. |
| Formulation & Reconstitution | PBS liquid / Lyophilized from GM's Protein Stability Buffer2 (PSB2,Confidential Ingredients) or PBS (pH7.4); For PSB2, reconstituted with 0.9% sodium chloride; For PBS, reconstituted with ddH2O. |
| Storage | Store at -20℃ to -80℃ under sterile conditions. Avoid repeated freeze-thaw cycles. |
| Cat No. of Products | GMP-h-fFN-Ab01, GMP-h-fFN-Ab02, GMP-h-fFN-Ab03, GMP-h-fFN-Ab04, GMP-h-fFN-Ab05 |
| Product Name | Anti-human FN1/fFN/FN mouse monoclonal antibody (mAb) |
| Target/Biomarker | Fetal fibronectin (fFN) |
| Alias of Target/Biomarker | CIG, ED-B, FINC, FN, FNZ, GFND, GFND2, LETS, MSF, SMDCF |
| Expression platform | CHO |
| Bioactivity validation | Human Fetal fibronectin (fFN) antigen binding, ELISA validated as capture antibody and detection antibody. Pair recommendation with other Fetal fibronectin (fFN) antibodies in fFN level test of Fertility (premature delivery) and related syndrome evaluation. |
| Tag | mFc |
| Products description | Anti-human FN1/fFN/FN mouse monoclonal antibody (mAb) is a monoclonal antibody produced by CHO. The antibody is ELISA validated as capture antibody and detection antibody pair. |
| Purity | Purity: ≥95% (SDS-PAGE) |
| Application | ELISA test, Lateral flow immunoassay (LFIA), colloidal gold immunochromatographic assay, Chemiluminescent immunoassay (CLIA), turbidimetric inhibition immuno assay (TINIA), immunonephelometry and POCT |
| Formulation & Reconstitution | Lyophilized from GM's Protein Stability Buffer2 (PSB2,Confidential Ingredients) or PBS (pH7.4); For PSB2, reconstituted with 0.9% sodium chloride; For PBS, reconstituted with ddH2O. |
| Storage | Store at -20℃ to -80℃ under sterile conditions. Avoid repeated freeze-thaw cycles. |
Reference
Validation Data
GeneMedi provides 5 kinds of Anti-human fFN mouse monoclonal antibody(mAb): GMP-h-fFN-Ab01; GMP-h-fFN-Ab02; GMP-h-fFN-Ab03; GMP-h-fFN-Ab04; GMP-h-fFN-Ab05. Our R&D department demonstrated that our Anti-human fFN mouse monoclonal antibody pair has a large linear range and good sensitivity against the GMP-h-fFN-Ag01. Below is the result of GeneMedi's GMP-h-fFN-Ab pair validation with GMP-h-fFN-Ag01 in ELISA. We highly recommend the Ab&Ag to you.
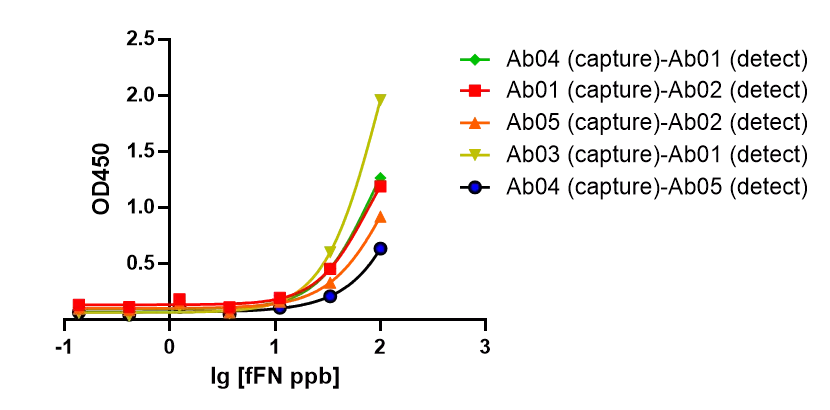
Figure 1: GeneMedi's GMP-h-fFN-Ab pairs are validated to detect the GMP-h-fFN-Ag01 in ELISA.
EC50 [Ab01 (capture)-Ab02 (detect)]= 76.68 ppb;
EC50 [Ab05 (capture)-Ab02 (detect) ]= 130.7 ppb;
EC50 [Ab03 (capture)-Ab01 (detect) ]= 97.78 ppb;
EC50 [Ab04 (capture)-Ab01 (detect) ]= 79.94 ppb;
EC50 [Ab04 (capture)-Ab05 (detect) ]= 530.8 ppb.
Click to get more Data / Case study about the product.
Target/Biomarker information
Fetal fibronectin (fFN) is a protein produced at the boundary between the amniotic sac (which surrounds the baby) and the lining of the mother’s uterus (the decidua). Fetal fibronectin is largely confined to this junction and is thought to help “glue” or maintain the integrity of the boundary between the amniotic sac and the lining of the uterus. A fetal fibronectin test detects fFN in vaginal fluid to help predict the short-term risk of premature delivery. According to the American College of Obstetricians and Gynecologists (ACOG), a normal pregnancy is about 40 weeks, with a woman going into labor between 37 and 42 weeks. A preterm delivery is one that occurs between 20 and 37 weeks of pregnancy. fFN is found in vaginal fluids early in pregnancy because of the normal growth and establishment of tissues at the junction between the amniotic sac and uterus, with levels falling when this phase is complete. fFN is also found in fluids from the vagina after 36 weeks when it is often released into vaginal fluids as the body gets ready for childbirth. However, fFN should not be detectible between 22 and 35 weeks of pregnancy. Elevated levels during this period reflect a disturbance at the junction between the amniotic sac and the lining of the uterus. Elevated fFN in vaginal fluids during these weeks of pregnancy has been associated with an increased risk of preterm labor and delivery. Many pregnant women experience symptoms that suggest preterm labor. These may include uterine contractions, changes in vaginal discharge, backaches, abdominal discomfort, pelvic pressure, cramping, and dilation of the cervix. However, not all symptomatic women will actually have a preterm delivery. The majority will go on to deliver at term. Unfortunately, while premature births can have successful endings, serious complications are possible when a baby leaves the womb early. Premature babies frequently have difficulty breathing and feeding. Their lungs and other organs are immature and do not function normally, and the strain on them can cause persistent health problems. The more premature the newborn, the more likely it is that he or she will experience complications. If a healthcare practitioner thinks that a woman might deliver early, she will consider treatments designed to delay delivery. These treatments can have unwanted side effects, however, so knowing whether or not a woman is likely to deliver prematurely helps in the decision on the best course of action. The fFN test is a relatively noninvasive tool that can help distinguish between those women who are likely to deliver shortly and those who are not.
About GDU

GDU helps global diagnostic partners in high quality of raw material discovery, development, and application. GDU believes in Protein&antibody Innovation for more reliable diagnostic solutions.






 Go to Fertility diagnostics products collection
>>
Go to Fertility diagnostics products collection
>>
Comments