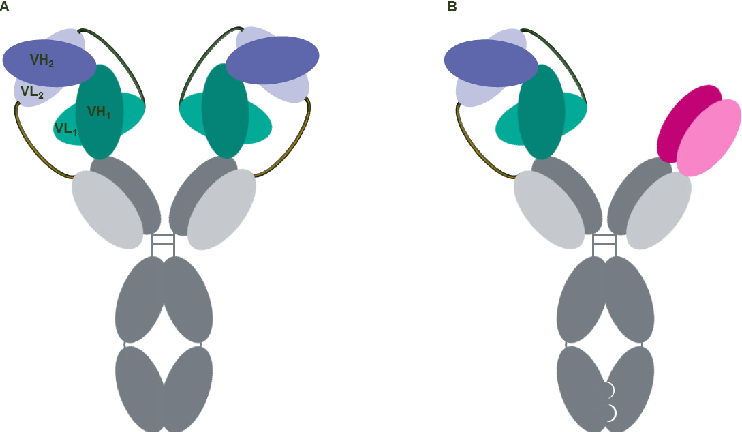
CODV (cross-over dual variable domains) -Ig, contain four polypeptide chains that form two dual variable domains (four antigen binding sites) with a cross-over orientation (Figure 1), which is attained by inverting the alignment of the cognate domains on one chain. In order to adopt the correct VH/VL pairing, linker combinations were designed and optimized using a molecular modeling strategy. The overall CODV structure replicates a circular self-contained architecture (Figure 1A), with binding sites facing up to opposite sites and able to accommodate a large variety of antigen sizes while maintaining parental affinities. The CODV structure was also further developed in a trispecific format in which a single IgG Fab arm is combined with a double arm generated in the CODV structure using the KiH heterodimerization strategy (Figure 1B). A trispecific CODV molecule was successfully engineered to target three distinct epitopes on human immunodeficiency virus HIV-1 envelope, including the CD4 binding site, MPER and the V1V2 glycan site. This innovative molecule exhibited an unprecedented neutralization breadth and potency against HIV when compared to other previously described broadly neutralizing antibodies (BnAbs).