Adeno-Associated Virus(AAV) Gene Therapy Landscape
Adeno-associated viruses (AAV) are virus particles composed of single-stranded DNA surrounded by a protein shell. Despite their simple structure, recombinant AAVs (rAAV) can perform the important role of delivering nucleic acids into cells during gene therapy. Due to the high demand for potentially curative treatments in areas of huge unmet need and the growing availability of AAV vectors, a race to launch successful gene therapy products is underway. Companies such as Novartis and Spark Therapeutics already have approved gene therapy products and the field is growing, with at least 50 companies/institutions actively developing AAV gene therapy products (Figure 1 and Table 1).
Figure 1: Companies developing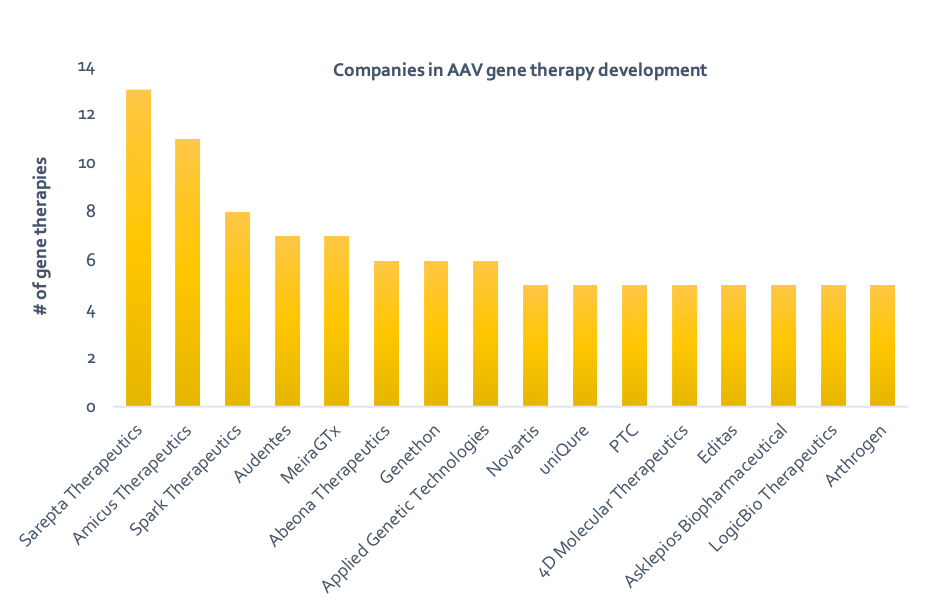 Table 1: Companies developing
Table 1: Companies developing 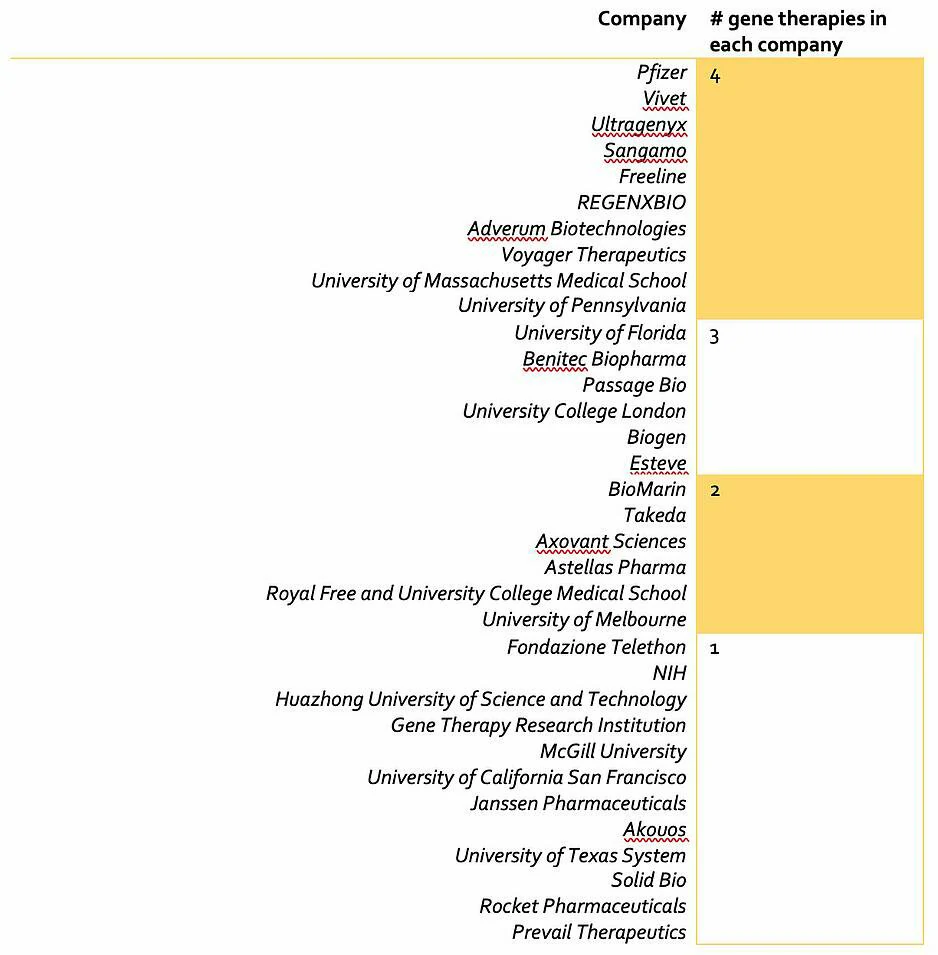 Advantages of using AAVs for gene therapy include their ability to infect both non-dividing and dividing cells, lack of pathogenicity and relatively poor immunogenicity. In addition, AAVs act episomally by behaving as separate extrachromosomal elements in the nucleus of target cells – this decreases their propensity to cause cancer. However, AAVs are not without their disadvantages. One limitation is their nucleic acid capacity, with AAVs having a genome size 5-10x smaller than adenoviruses or lentiviruses. Another non-trivial challenge facing effective and safe AAV gene delivery is the host immune response.
Advantages of using AAVs for gene therapy include their ability to infect both non-dividing and dividing cells, lack of pathogenicity and relatively poor immunogenicity. In addition, AAVs act episomally by behaving as separate extrachromosomal elements in the nucleus of target cells – this decreases their propensity to cause cancer. However, AAVs are not without their disadvantages. One limitation is their nucleic acid capacity, with AAVs having a genome size 5-10x smaller than adenoviruses or lentiviruses. Another non-trivial challenge facing effective and safe AAV gene delivery is the host immune response.
The Immune Response
The humoral immune response is particularly problematic for AAV gene therapy, with 96% of humans having antibodies against AAV, of which 32% neutralize AAVs completely (Chirmule et al., 1999). The presence of neutralizing antibodies is one of the major factors that led to early clinical failures in this approach (Greenberg et al., 2015). The issue is further complicated by site-specific immune responses to different AAV serotypes; hence when delivering an AAV based gene therapy, an understanding of the best route of administration for a specific AAV is required (Brockstedt et al., 1999). There are four main strategies that can be adopted to address the presence of anti-AAV neutralizing antibodies. The first strategy involves using less-seroprevalent capsids or switching serotype. Boutin and colleagues demonstrated that the prevalence of anti-AAV1 and -AAV2 total IgG is higher (67% and 72% respectively) than that of anti-AAV5 (40%), anti-AAV6 (46%), anti-AAV8 (38%) and anti-AAV9 (47%) (Boutin et al., 2010). Serotype switching has been shown to be successful but is only a short-term solution that circumvents the challenge posed by anti-AAV neutralizing antibodies. A second option would be to perform plasmapheresis. Plasmapheresis is the process by which neutralizing antibodies can be filtered from patient blood. This method has proven successful in reducing the titers of neutralizing factors for AAV types 1,2,6, and 8 in a sample of ten human patients (Monteilhet et al., 2011). In addition to addressing issues with primary infection with AAVs, this method can also be applied to patients requiring re-administration of vector. Limitations include the fact that the method requires multiple cycles of plasmapheresis and has been shown to be less efficient in patients with high titers of neutralizing antibodies. A third suggestion has been to use immunosuppressants. This method appears to be the preferred short-term solution, primarily due to the ease of access to low cost immunosuppressive drugs. The potential of immunosuppressants in addressing the problem was shown by Velazquez and colleagues (Velazquez et al., 2017). The research group found that combination administration of prednisolone and rapamycin reduced serum AAV9 neutralizing antibodies by 70-80% at 4 weeks and 85%-93% at 8 weeks of treatment. One major issue with using immunosuppressants is that it makes the user vulnerable to infections. These drugs may also have unwanted consequences on tissue biodistribution and transduction efficiency of the gene therapy. A fourth option is to increase the capsid dose or to use capsid decoys. Mingozzi et al. demonstrated that introducing empty capsids along with the gene therapy vector would titrate out any neutralizing antibodies to AAV - these empty capsids served as a decoy to allow successful transgene delivery (Mingozzi et al, 2013). However, this method has yet to be tested in humans and is likely to be ineffective with high antibody titers. Furthermore, as found by Johnson and Samulski, empty capsids would also enter target cells, leading to an increased presentation of capsid epitopes on MHC class I molecules (Johnson and Samulski, 2008). This would in theory increase the harmful T cell responses against the AAV gene therapy capsid. Clearly the immune system response against the AAV capsid still poses the biggest challenge for successful AAV gene therapy. Despite this, companies continue to develop gene therapies using a wide variety of serotypes for many indications.Gene Therapy Landscape by AAV Serotype
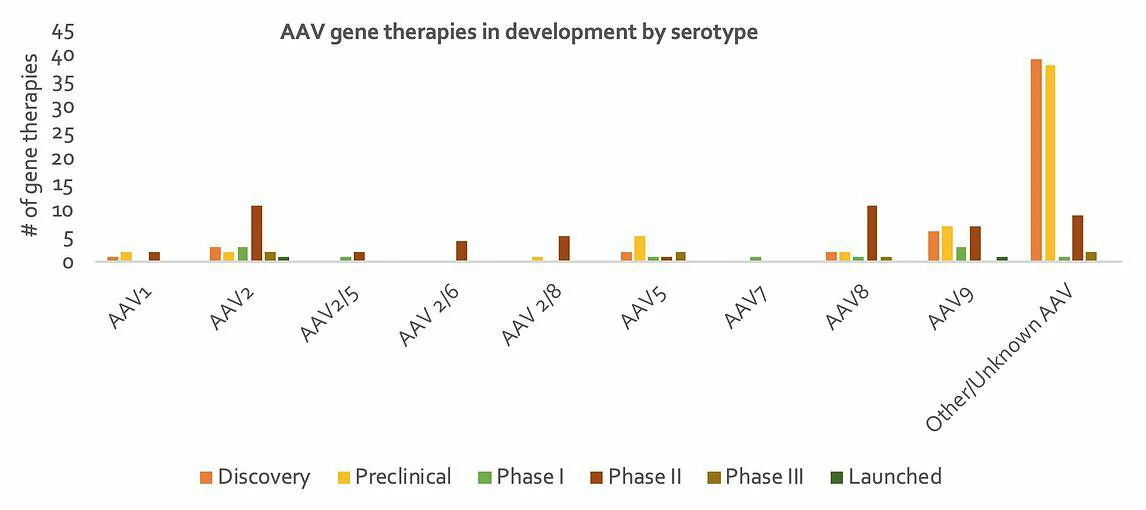
At present, 12 human serotypes and more than 100 non-human primate serotypes have been identified. A landscaping exercise of companies developing gene therapy products (see Figure 2) revealed that while the specific serotypes utilized in many programs remain undisclosed, largely due to their early stage in development, AAV2, AAV8 and AAV9 are the most popular serotypes being utilized amongst those programs for which AAV serotype is publicly known. There are of course many factors that contribute to serotype selection, one of which is tissue tropism. Figure 2: (A) Number of companies developing in-vivo gene therapy products, by AAV serotype (serotype, # of companies); (B) Number of in-vivo gene therapy products in development, by AAV serotype (serotype, # of products).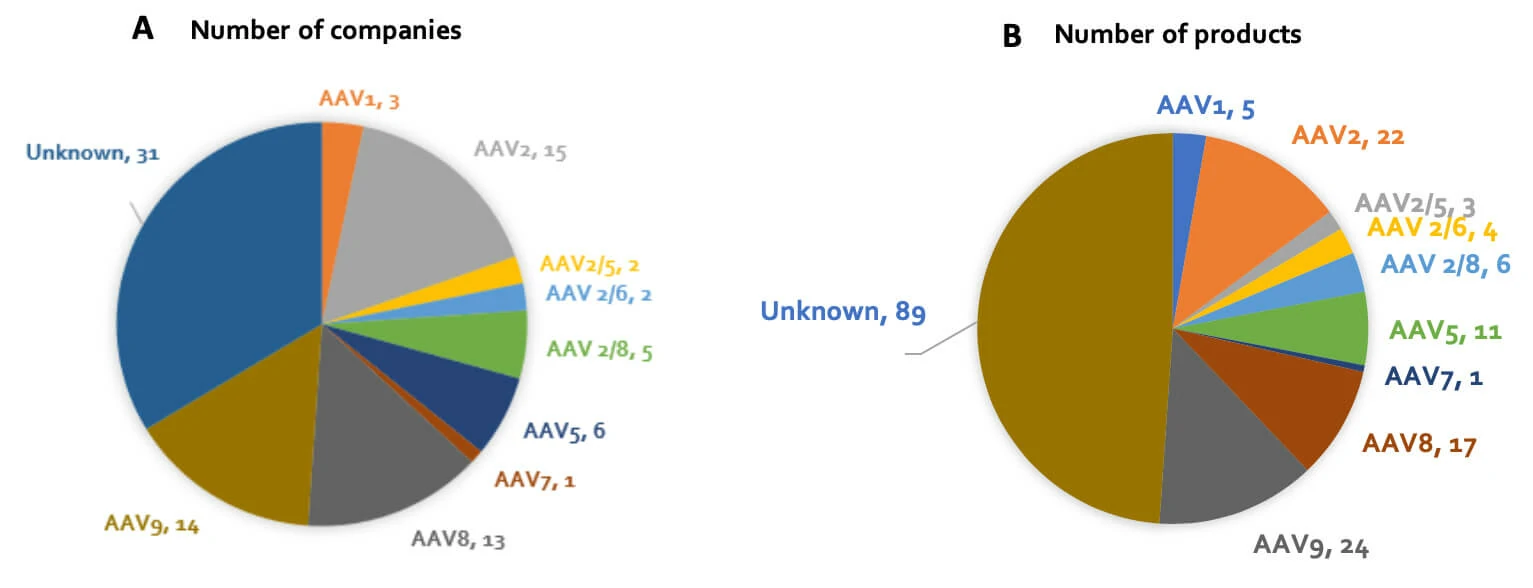 AAV serotypes differ in their tropism i.e. their ability to infect certain cell types, principally due to their different cell surface receptors. Our current understanding of inherent AAV tropism is outlined in Table 2. Following the discovery that cell surface proteins influence AAV tropism, researchers have been able to genetically modify surface proteins to target certain AAVs to specific cell types. For example, investigators (Ried et al., 2002) have incorporated ankyrin repeat proteins (DARPins, sections of protein A) and cytokines into an AAV capsid for specific cell targeting. This method has been subsequently applied to create AAVs targeting tumors and CD4+ T cells, with implications for cancer treatment (Münch et al., 2013). As we continue develop our understanding of AAV biology, we will undoubtedly find more opportunities to modify the properties of AAVs to better perform their function as gene therapeutics.
Table 2: Tissue tropisms by AAV serotype (AddGene)
AAV serotypes differ in their tropism i.e. their ability to infect certain cell types, principally due to their different cell surface receptors. Our current understanding of inherent AAV tropism is outlined in Table 2. Following the discovery that cell surface proteins influence AAV tropism, researchers have been able to genetically modify surface proteins to target certain AAVs to specific cell types. For example, investigators (Ried et al., 2002) have incorporated ankyrin repeat proteins (DARPins, sections of protein A) and cytokines into an AAV capsid for specific cell targeting. This method has been subsequently applied to create AAVs targeting tumors and CD4+ T cells, with implications for cancer treatment (Münch et al., 2013). As we continue develop our understanding of AAV biology, we will undoubtedly find more opportunities to modify the properties of AAVs to better perform their function as gene therapeutics.
Table 2: Tissue tropisms by AAV serotype (AddGene)
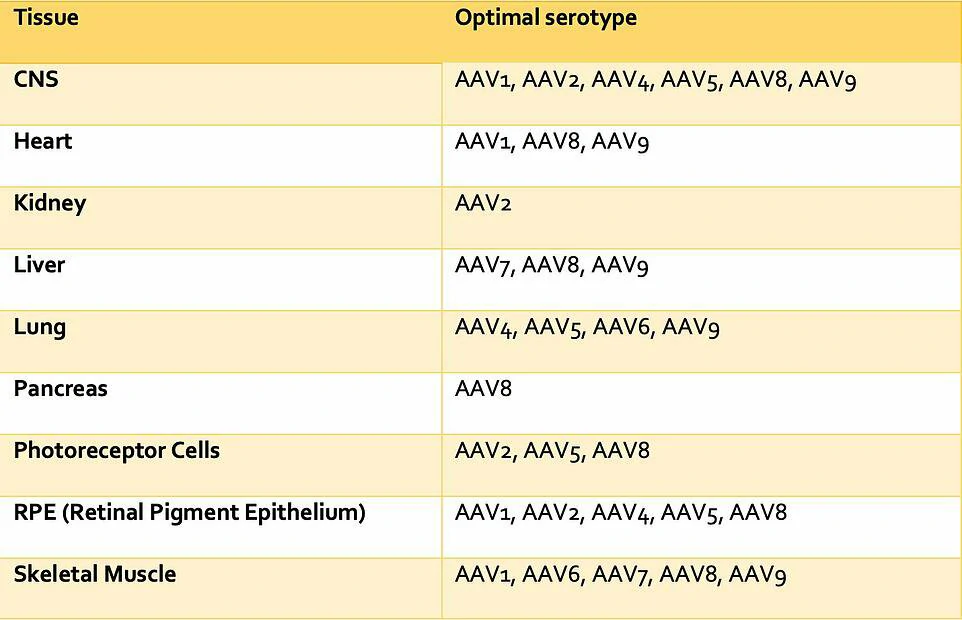 Figure 3: Number of in-vivo AAV gene therapy products under different stages of development, by serotype.
Figure 3: Number of in-vivo AAV gene therapy products under different stages of development, by serotype.
 AAV2 was one of the first AAVs to be identified and is one of the serotypes for which we have the most detailed understanding. It is therefore no surprise that a large proportion of late clinical stage gene therapy products utilize AAV2, as can be seen in Figure 3. Many gene therapies also utilize AAV8 or AAV9 due to their well-established tropism for the liver, heart and skeletal muscle following intravascular delivery. For example, Solid Biosciences is using AAV9’s specificity for skeletal muscle in its treatment of Duchenne Muscular Dystrophy (DMD) (Phase II, SGT-001). AAV9’s efficiency in traversing the blood-brain barrier has also brought new opportunities for the treatment of CNS disorders, including those involving non-neuronal cells. An example of such an application is Amicus Therapeutics’ gene therapy to target neurons for the treatment of Batten Disease (Phase I/II, CLN3/CLN6). Gene therapies utilizing AAV5 are also being developed centred around its tropism. For example, Editas Medicine is using AAV5’s specificity for retinal photoreceptors for the treatment of LCA (Phase 2, EDIT-101).
AAV2 was one of the first AAVs to be identified and is one of the serotypes for which we have the most detailed understanding. It is therefore no surprise that a large proportion of late clinical stage gene therapy products utilize AAV2, as can be seen in Figure 3. Many gene therapies also utilize AAV8 or AAV9 due to their well-established tropism for the liver, heart and skeletal muscle following intravascular delivery. For example, Solid Biosciences is using AAV9’s specificity for skeletal muscle in its treatment of Duchenne Muscular Dystrophy (DMD) (Phase II, SGT-001). AAV9’s efficiency in traversing the blood-brain barrier has also brought new opportunities for the treatment of CNS disorders, including those involving non-neuronal cells. An example of such an application is Amicus Therapeutics’ gene therapy to target neurons for the treatment of Batten Disease (Phase I/II, CLN3/CLN6). Gene therapies utilizing AAV5 are also being developed centred around its tropism. For example, Editas Medicine is using AAV5’s specificity for retinal photoreceptors for the treatment of LCA (Phase 2, EDIT-101).
Disease Areas Being Targeted by In-Vivo Gene Therapy
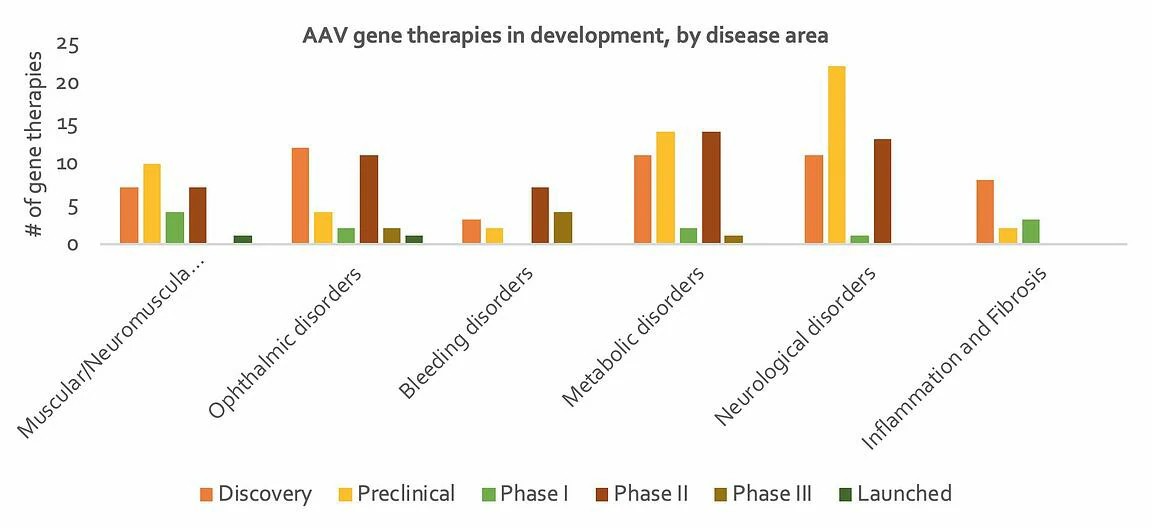
At present, many companies are focusing their efforts on developing gene therapies to treat neurological, ophthalmic and metabolic disorders, for which many indications result from loss-of-function genetic alterations (Figure 4). The eye’s immune-privileged status, accessibility and compartmentalization significantly reduces systemic spread of locally delivered gene therapy product and therefore reduces the risk of antibody neutralization of the vector. Plus, manufacturing costs are comparatively low given the small relative size of the eye. The liver is also an important target for gene therapy; many metabolic and bleeding disorders are a result of dysfunctional enzymes expressed predominantly or exclusively in the liver. Because of the liver’s unique dual blood supply, it receives nearly 25% of the cardiac output of blood. This means that administration of an AAV systemically leads to the effective delivery of the vector to hepatocytes. The use of AAVs in muscular disorders is also promising due to their high transgene expression when injected intramuscularly, as well as whole-body muscle transduction when delivered intravenously. Furthermore, skeletal muscle cells do not divide after formation, which helps minimise the loss of transgene expression. Figure 4: (A) Number of companies developing an in-vivo AAV gene therapy product, by disease area; (B) Number of in-vivo AAV gene therapy products being developed, by disease area.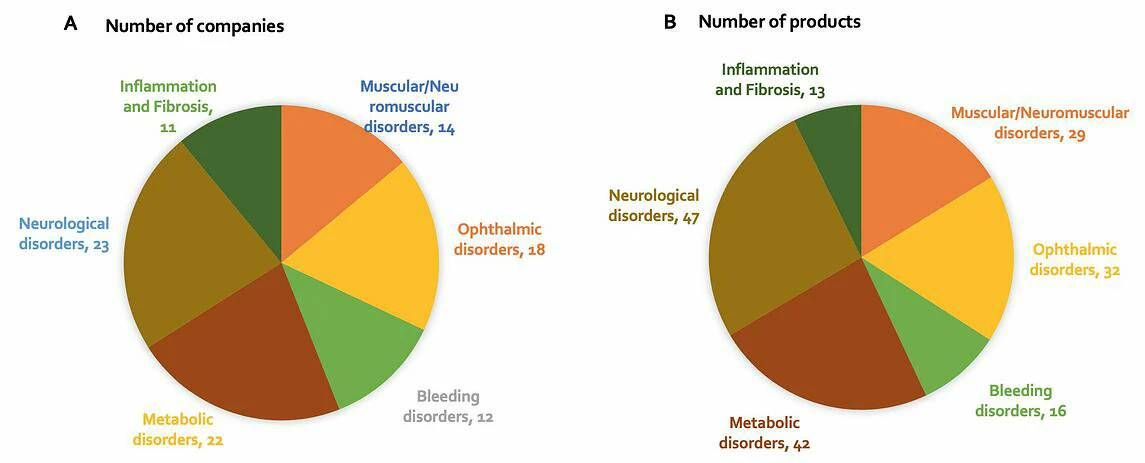 A review of current gene therapies in development by disease area and stage of development shows that while activity is relatively evenly spread, there are notably more gene therapies in preclinical stage for neurological disorders than other disease areas, suggesting less progression in this particular area. It has historically been difficult to pinpoint specific genes involved in disease in this area, and of course delivery to the brain is difficult. On the other hand, bleeding disorders account for the highest number of gene therapies in Phase 3 (see Figure 5). Gene therapies for Haemophilia make up 13 out of the 16 products addressing bleeding disorders; there is a large unmet need for treatments in this indication, given that the current standard of treatment, clotting factor replacement, must be administered for the life of the patient. It is therefore no surprise that so many gene therapy products for this indication are progressing rapidly through development, especially those that demonstrate long-term duration. Interestingly, gene therapies actively in development for inflammation / fibrosis have only progressed to Phase 1. Four out of the 13 products addressing inflammation / fibrosis disorders are for treating cystic fibrosis. To date, one of the major challenges in developing gene therapies for this indication has been to fit the CFTR gene, alongside a suitably strong promoter, into a limited capacity AAV. Furthermore, it is notoriously difficult to deliver a vector to target cells in the lung given its highly protected status, and expression of the CFTR gene is quickly lost due to high turnover rates of airway cells.
Figure 5: Number of in-vivo AAV gene therapy products under different stages of development, by disease area
A review of current gene therapies in development by disease area and stage of development shows that while activity is relatively evenly spread, there are notably more gene therapies in preclinical stage for neurological disorders than other disease areas, suggesting less progression in this particular area. It has historically been difficult to pinpoint specific genes involved in disease in this area, and of course delivery to the brain is difficult. On the other hand, bleeding disorders account for the highest number of gene therapies in Phase 3 (see Figure 5). Gene therapies for Haemophilia make up 13 out of the 16 products addressing bleeding disorders; there is a large unmet need for treatments in this indication, given that the current standard of treatment, clotting factor replacement, must be administered for the life of the patient. It is therefore no surprise that so many gene therapy products for this indication are progressing rapidly through development, especially those that demonstrate long-term duration. Interestingly, gene therapies actively in development for inflammation / fibrosis have only progressed to Phase 1. Four out of the 13 products addressing inflammation / fibrosis disorders are for treating cystic fibrosis. To date, one of the major challenges in developing gene therapies for this indication has been to fit the CFTR gene, alongside a suitably strong promoter, into a limited capacity AAV. Furthermore, it is notoriously difficult to deliver a vector to target cells in the lung given its highly protected status, and expression of the CFTR gene is quickly lost due to high turnover rates of airway cells.
Figure 5: Number of in-vivo AAV gene therapy products under different stages of development, by disease area