CAR-NK immunotherapy and cancer therapy
 Lentivirus Protocol Download
Lentivirus Protocol Download
Although CAR-T has been applied in clinic, quite a lot of side effects, such as off-target, cytokine release syndrome (CRS) etc., pose a great restriction in their further clinical application [47]. Based on natural killer (NK) cells, CAR-NK cell therapy is much safer than CAR-T therapy and becomes a good candidate for targeting and combating solid tumors [48,49]. Based on the four generation of CAR, CARs in CAR-NK are designed with the addition of personalized proteins, such as DNAX-activation protein (DAP) 12 and NKG2 member D (NKG2D), which show great anti-tumor activity in acute myeloid leukemia (ALL), osteosarcoma, and prostate tumors [50-52]. Therefore, CAR-NK cells not only have the ability of CAR to specifically recognize antigen-expressing tumors, but also can destroy tumors via NK cell receptors. To date, numerous CAR-NK cell therapies have been used for anti-tumor therapy in clinical trials (Table 3).
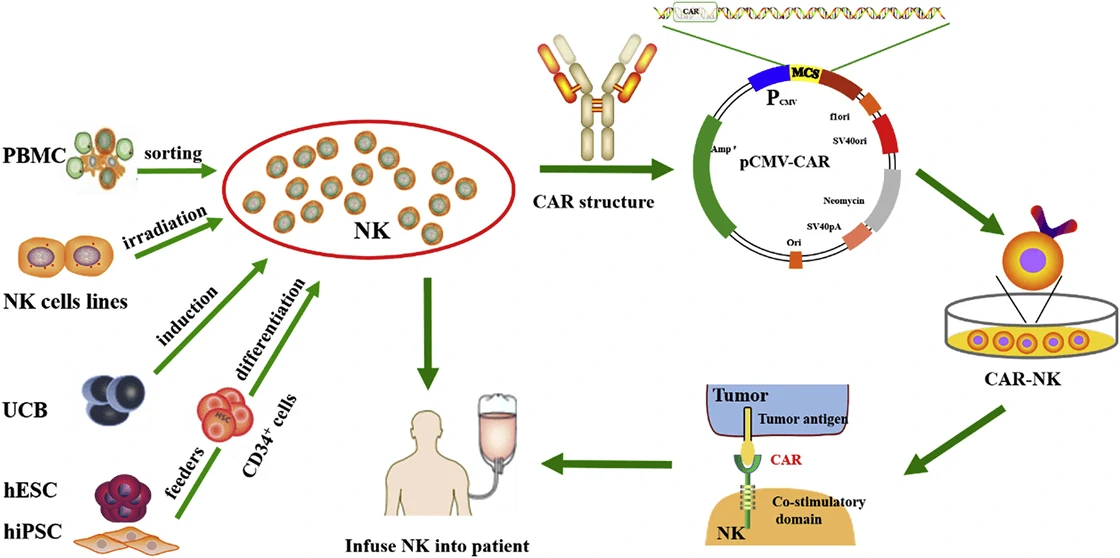 Figure 4 Schematic of CAR-NK cell therapy [53]. Gene engineered natural killer (NK) cells are infused into patients to destroy tumor cells. PBMC, peripheral blood mononuclear cell; UCB, umbilical cord blood; ESCs, human embryonic stem cells; iPSCs, human induced pluripotent stem cells.
Figure 4 Schematic of CAR-NK cell therapy [53]. Gene engineered natural killer (NK) cells are infused into patients to destroy tumor cells. PBMC, peripheral blood mononuclear cell; UCB, umbilical cord blood; ESCs, human embryonic stem cells; iPSCs, human induced pluripotent stem cells.
| Antigen | Disease | NK source | Phase of clinical trials | NCT/Reference |
| CD7 | Lymphoma,leukemia, | NK-92 | Phase I/II | NCT02742727 |
| CD19 | Lymphoma,leukemia, | NK-92 | Phase I/II | NCT02892695 |
| CD33 | Acute myeloid leukemia | NK-92 | Phase I/II (complete) | NCT02944162 |
| MUC1 | Solid tumors | NK-92 | Phase I/II | NCT02839954 |
| NR | Non-small cell lung carcinoma | NK-92 | Phase I | NCT03656705 |
| HER2 | Glioblastoma multiforme | NK-92 | Phase I | NCT03383978 |
| CD19 | B-acute lymphocytic leukaemia | PB-NK | Phase II | NCT01974479 |
| CD19 | B-acute lymphocytic leukaemia | PB-NK | Phase I (complete) | NCT00995137 |
| CD19 | B-lymphoma | UCB-NK | Phase I/II | NCT03056339 |
View Knowledge Base - Landscape of Advanced cell therapy>>




