Lentivirus Vector System (Lentivurs packaging and expression system) | GeneMedi
Order Information
Introduction to Lentivirus Vector System (Lentivurs packaging and expression system)
Lentivirus are based on HIV-1 (human immunodeficiency virus type I), which has been one of the most widely used gene therapy vectors. As a powerful tool for introduction of exogenous genes, there are many advantageous features of lentivirus vectors, such as mediating efficient transfection and long-term expression of exogenous genes in both dividing and non-dividing cells. To date, lentivirus vector has been widespread utilized in various cell lines for gene overexpression, RNA interference, microRNA research and in vivo animal experiments.
A lentivirus packaging system including the lentivirus expression vector, the lentivirus package plasmid and helper plasmid. In order to meet different infection needs, Genemedi has launched a variety of lentivirus packaging system plasmids with different fluorescent labels (such as GFP, RFP, mCherry and so on), and various promoters or tags, which may also allow researchers to produce lentiviruses by themselves as they need.
Properties
| Genemedi Lentivirus Vector System Plasmids | |
|---|---|
| Quantity/Unit | vials |
| Form | Liquid |
| Sipping and Storage Guidelines | Shipped at 4 ° C and stored at -20 ° C, effective for 1 year. |
| Titer | > 5ug each |
Advantages
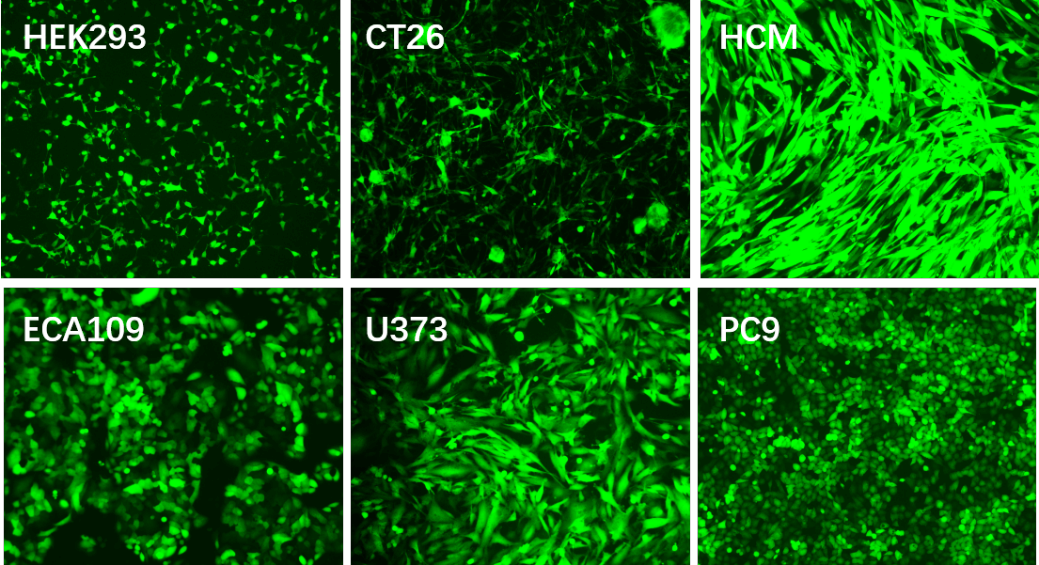
Applications and Figures
Technical Documents
 Lentivirus User Manual.
Lentivirus User Manual.Frequently Asked Questions(FAQs)
- 1. What is the difference between 2nd generation and 3rd generation lentiviral systems?
-
AnswerBriefly, 2nd generation lentiviral systems use more HIV proteins (on fewer plasmids) in order to produce functional lentiviral particles than 3rd generation systems. 2nd generation packaging systems: express the HIV gag, pol, rev, and tat genes all from a single packaging plasmid such as psPAX2. 3rd generation packaging systems: express gag and pol from one packaging plasmid and rev from another, such aspMDLg/pRRE and pRSV-Rev. 3rd generation packaging systems do not express tat. Third generation lentiviral systems are considered safer than second generation systems, but may be more difficult to use, because they require transfection with four separate plasmids in order to create functional lentiviral particles.
- 2. What is the difference between lentivirus and retrovirus?
-
AnswerLentiviruses are a subtype of retrovirus. From an experimental standpoint, the main difference between lentiviruses and standard retroviruses (γ-retroviruses) is that lentiviruses are capable of infecting non-dividing and actively dividing cell types whereas standard retroviruses can only infect mitotically active cell types. This means that lentiviruses can infect a greater variety of cell types than retroviruses. Both lentiviruses and standard retroviruses use the gag, pol, and env genes for packaging; however, they are different viruses and thus use slightly different isoforms of these packaging components. Therefore, lentivirus may not be efficiently packaged by retroviral packaging systems, and vice versa.
- 3. Which bacterial strain should be used for cloning and producing my lentiviral plasmids?
-
AnswerDue to the long terminal repeats found in lentiviral plasmids, we recommend using a strain that reduces the frequency of homologous recombination of unstable regions, such as Invitrogen Stbl3™ or NEB Stable cells. This will ensure that the repeats will be maintained and often results in a greater yield of DNA. However, if the plasmid contains a Gateway cassette containing the ccdB gene, a ccdB survival strain is necessary.
- 4. Which cell line should be used to produce lentivirus?
-
Answer293T cells are usually used to produce lentivirus.
- 5. What dictates lentiviral host cell range (tropism)?
-
AnswerLentiviral tropism is determined by the ability of the viral envelope protein to interact with receptors at the host cell surface. The VSV-G envelope protein is commonly used in lentiviral particle production because it confers broad tropism over a range of species and cell types. For more information, see the Cronin, et al. article on different envelopes and their tropism.
- 6. How can lentivirus be used to make stable cell lines?
-
AnswerLentiviruses can be used to make stable cell lines in the same manner as standard retroviruses. That is, many lentiviral genomes have selectable markers, such as the puromycin resistance gene, conferring antibiotic resistance to infected host cells. When these antibiotics are added to the growth medium of the host cells, they kill off any cells that have not incorporated the lentiviral genome and those cells that survive can be expanded to create stable cell lines, which have incorporated the lentiviral genome and harbor the genetic information encoded by that genome. Many lentiviral transfer plasmids do not have selectable markers conferring resistance to an antibiotic, but do encode another marker, such as GFP. A researcher can use FACS to sort cells expressing GFP and later expand these cells into a cell line.
- 7. Where does lentivirus integrate?
-
AnswerGenome-wide studies of viral integration have shown that lentiviruses most often integrate into actively transcribed genes, and that this preference is conserved across target species. Although chromatin availability facilitates integration, it does not explain the lentiviral preference for transcribed genes. Studies comparing the lentivirus HIV and the retrovirus MMLV indicate that the viral integrase plays a role in shaping integration site preferences. A major cellular determinant is LEDGF/p75, a lentiviral tethering protein that recruits the pre-integration complex to transcriptional units and facilitates integration. LEDGF/p75 binding sites are enriched in gene bodies and mostly absent in promoters and intergenic regions, mirroring patterns of lentiviral integration.
- 8. Can lentiviral plasmids be used in direct transfections as opposed to making virus?
-
AnswerSome (but not all) lentiviral transfer plasmids can be used in transient transfections to achieve expression of the transgene, and those that can are primarily third generation constructs. Lentiviral transfer plasmids are not designed specifically for transient transfections. Therefore, there may be limited transgene expression due to the lentiviral LTRs. While possible, it is not explicitly recommended that you use lentiviral transfer plasmids for simple transfections.
- 9. Is it feasible to express cDNA from a lentiviral plasmid normally used for shRNA expression?
-
AnswerYes, it is feasible, but first the promoter within the transfer plasmid must be modified. Most shRNA‐expressing lentiviral plasmids, such as pLKO.1, use a U6 or H1 promoter in order to drive RNA pol III-directed transcription of shRNAs. cDNA expression requires RNA pol II, and thus requires an RNA pol II promoter, such as CMV or RSV.
- 10. What techniques can be used to clone an insert into a lentiviral plasmid containing only one restriction site?
-
AnswerIf a lentiviral transfer plasmid contains a single restriction site, one can use standard cloning techniques to ligate the insert into this site. If it is not immediately feasible to digest and clone the insert from a parent vector, some possible approaches to using this site include subcloning or appending compatible restriction sites onto the insert of interest using PCR. The process of subcloning consists of digesting the insert of interest from its parent vector and ligating into a second vector in such as way that the insert may later be digested from this new vector and cloned into the lentiviral vector. This is basically shuffling restriction sites between vectors until the gene of interest is flanked by sites compatible with those in the vector into which one ultimately wants to ligate the insert. Often times it is less time consuming and easier to simply add restriction sites onto the insert of interest using PCR. This is accomplished by PCR amplifying the insert sequence using primers that contain the restriction sites needed. Functional restriction sites must be a certain number of bases from the ends of the primers used. Alternatively, you could ligate a multiple cloning site (MCS) from a separate vector into the single site in the lentiviral vector and generate more useful restriction sites.
- 11. What safety concerns surround the use of lentiviral vectors?
-
AnswerAs noted by the NIH, the two main safety concerns surrounding the use of lentiviral are:
1. The potential for generation of replication-competent lentivirus
2. The potential for oncogenesis
The potential for generation of replication-competent lentivirus is addressed by the design of the vectors and by safe laboratory practice. In terms of vector design, 2nd and 3rd generation lentiviral systems provided by Addgene separate transfer, envelope, and packaging components of the virus onto different vectors. The transfer vector encodes the gene of interest and contains the sequences that will incorporate into the host cell genome, but cannot produce functional viral particles without the genes encoded in the envelope and packaging vectors. Unless recombination occurs between the packaging, envelope, and transfer vectors, and the resulting construct is packaged into a viral particle, it is not possible for viruses normally produced from these systems to replicate and produce more virus after the initial infection. In this regard, 3rd generation systems are considered safer than 2nd generation systems because the packaging vector has been divided into two separate plasmids (resulting in a four plasmid system in total). In addition, 3rd generation systems do not use the HIV protein tat in order to produce full length virus from the transfer vector during the viral production stage.
Many of the lentiviral transfer vectors that have been deposited with Hanbio are self-inactivating (SIN) vectors. These vectors have a deletion in the 3'LTR of the viral genome that is transferred into the 5'LTR after one round of reverse transcription. This deletion abolishes transcription of the full-length virus after it has incorporated into a host cell.
The potential for oncogenesis is largely based on the specific insert contained within the lentiviral transfer vector (dependent upon whether or not it is an oncogene) and should be considered on a case by case basis.
Biosafety should always be considered with respect to the precise nature of experiments being performed, and your biosafety office can provide more information on your institution's best practices with regard to lentiviral research. - 12. How can I increase the titer of my lentivirus?
-
AnswerHere are some suggestions for higher-titer lentivirus production:
1. Because lentivirus contains LTR repeat sequences, recombination can happen after transformation, resulting in a plasmid of reduced size. We recommend using Invitrogen Stbl3 competent cells for plasmid amplification, which will minimize recombination. If you did not use these cell lines to amplify your plasmids, you should confirm that the plasmids have maintained the correct size by running a restriction digest.
2. The insert should not exceed the cloning capacity of the expression vector. Viral packaging becomes less efficient with large inserts, so you can expect a low titer if you clone an insert size that approaches the maximum.
3. Transfection conditions should be optimized to achieve at least 80% transfection efficiency. Less than 80% may result in low titer.
4. Unlike VSVG pseudotyped virus, ecotropic viruses are not very stable, and can't handle ultracentrifugation or freeze/thaw cycles. If you are making ecotropic virus, it is best to infect target cells immediately upon harvesting the supernatant.
5.We recommend using 293T cells that are healthy and at a low passage number for packaging virus.
Reference
1. Wu, J. et al. MicroRNA-30 family members regulate calcium/calcineurin signaling in podocytes. Journal of Clinical Investigation 125, 4091-4106 (2015).
2. Li, F., Li, S. & Cheng, T. TGF-β1 Promotes Osteosarcoma Cell Migration and Invasion Through the miR- 143-Versican Pathway. Cellular Physiology and Biochemistry 34, 2169-2179 (2014).
3. Liu, Z. et al. miR-451a Inhibited Cell Proliferation and Enhanced Tamoxifen Sensitive in Breast Cancer via Macrophage Migration Inhibitory Factor. BioMed Research International 2015, 207684-207684 (2015).
4. Si, L. et al. Smad4 mediated BMP2 signal is essential for the regulation of GATA4 and Nkx2.5 by affecting the histone H3 acetylation in H9c2 cells. Biochemical and Biophysical Research Communications 450, 81-86 (2014).
5. Han, H., Yang, S., Lin, S. G., Xu, C. S. & Han, Z. Effects and mechanism of downregulation of COX‑2 expression by RNA interference on proliferation and apoptosis of human breast cancer MCF‑7 cells. Molecular Medicine Reports 10, 3092-3098 (2014).
6. Zhang, G., Liu, Z., Cui, G., Wang, X. & Yang, Z. MicroRNA-486-5p targeting PIM-1 suppresses cell proliferation in breast cancer cells. Tumor Biology 35, 11137-11145 (2014).
7. Li, G. et al. CYC1 silencing sensitizes osteosarcoma cells to TRAIL-induced apoptosis. Cellular Physiology and Biochemistry 34, 2070-2080 (2014).
8. Mao, J., Lv, Z. & Zhuang, Y. MicroRNA-23a is involved in tumor necrosis factor-α induced apoptosis in mesenchymal stem cells and myocardial infarction. Experimental and Molecular Pathology 97, 23-30 (2014).
9. Liu, X. et al. Role of human pulmonary fibroblast-derived MCP-1 in cell activation and migration in experimental silicosis. Toxicology and Applied Pharmacology 288, 152-160 (2015).
10. Guan, G. et al. CXCR4-targeted near-infrared imaging allows detection of orthotopic and metastatic human osteosarcoma in a mouse model. Scientific Reports 5, 15244-15244 (2015).
11. Zhang, Y. et al. Role of high-mobility group box 1 in methamphetamine-induced activation and migration of astrocytes. Journal of Neuroinflammation 12, 156-156 (2015).
12. Zhu, T. et al. The Role of MCPIP1 in Ischemia/Reperfusion Injury-Induced HUVEC Migration and Apoptosis. Cellular Physiology and Biochemistry 37, 577-591 (2015).
13. Qian, M. et al. P50-associated COX-2 extragenic RNA (PACER) overexpression promotes proliferation and metastasis of osteosarcoma cells by activating COX-2 gene. Tumor Biology 37, 3879-3886 (2016).
14. Wu, N., Song, Y., Pang, L. & Chen, Z. CRCT1 regulated by microRNA-520 g inhibits proliferation and induces apoptosis in esophageal squamous cell cancer. Tumor Biology 37, 8271-8279 (2016).
15. Wang, Y. et al. Overexpression of Hiwi Inhibits the Growth and Migration of Chronic Myeloid Leukemia Cells. Cell Biochemistry and Biophysics 73, 117-124 (2015).
16. Niu, L. et al. RNF43 Inhibits Cancer Cell Proliferation and Could be a Potential Prognostic Factor for Human Gastric Carcinoma. Cellular Physiology and Biochemistry 36, 1835-1846 (2015).
17. Zhang, H. et al. ZC3H12D attenuated inflammation responses by reducing mRNA stability of proinflammatory genes. Molecular Immunology 67, 206-212 (2015).
18. Deng, X. et al. MiR-146b-5p Promotes Metastasis and Induces Epithelial-Mesenchymal Transition in Thyroid Cancer by Targeting ZNRF3. Cellular Physiology and Biochemistry 35, 71-82 (2015).
19. Zhang, B. et al. HSF1 Relieves Amyloid-β-Induced Cardiomyocytes Apoptosis. Cell Biochemistry and Biophysics 72, 579-587 (2015).
20. Hu, Q. et al. Periostin Mediates TGF-β-Induced Epithelial Mesenchymal Transition in Prostate Cancer Cells. Cellular Physiology and Biochemistry 36, 799-809 (2015).
21. Yang, Z. et al. CD49f Acts as an Inflammation Sensor to Regulate Differentiation, Adhesion, and Migration of Human Mesenchymal Stem Cells. Stem Cells 33, 2798-2810 (2015).
22. Wang, X. et al. MCPIP1 Regulates Alveolar Macrophage Apoptosis and Pulmonary Fibroblast Activation After in vitro Exposure to Silica. Toxicological Sciences 151, 126-138 (2016).
23. Gu, S., Ran, S., Liu, B. & Liang, J. miR-152 induces human dental pulp stem cell senescence by inhibiting SIRT7 expression. FEBS Letters 590, 1123-1131 (2016).
24. Jin, F., Qiao, C., Luan, N. & Li, H. Lentivirus-mediated PHLDA2 overexpression inhibits trophoblast proliferation, migration and invasion, and induces apoptosis. International Journal of Molecular Medicine 37, 949-957 (2016).
25. Liu, Z., Song, Y., Wan, L., Zhang, Y. & Zhou, L. Over-expression of miR-451a can enhance the sensitivity of breast cancer cells to tamoxifen by regulating 14-3-3ζ, estrogen receptor α, and autophagy. Life Sciences 149, 104-113 (2016).
26. Tian, Y. et al. MicroRNA-30a promotes chondrogenic differentiation of mesenchymal stem cells through inhibiting Delta-like 4 expression. Life Sciences 148, 220-228 (2016).
27. Xu, S. et al. MicroRNA-33 promotes the replicative senescence of mouse embryonic fibroblasts by suppressing CDK6. Biochemical and Biophysical Research Communications 473, 1064-1070 (2016).
28. Chen, H., Sun, M., Liu, J., Tong, C. & Meng, T. Silencing of Paternally Expressed Gene 10 Inhibits Trophoblast Proliferation and Invasion. PLOS ONE 10 (2015).
29. Deng, Y. et al. Repair of critical-sized bone defects with anti-miR-31-expressing bone marrow stromal stem cells and poly(glycerol sebacate) scaffolds. European Cells & Materials 27, 13-25 (2014).
30. Zheng, Y. & Xu, Z. MicroRNA-22 induces endothelial progenitor cell senescence by targeting AKT3. Cellular Physiology and Biochemistry 34, 1547-1555 (2014).
31. Yang, X. et al. A lentiviral sponge for miRNA-21 diminishes aerobic glycolysis in bladder cancer T24 cells via the PTEN/PI3K/AKT/mTOR axis. Tumor Biology 36, 383-391 (2015).
32. Wang, W. et al. p53/PUMA expression in human pulmonary fibroblasts mediates cell activation and migration in silicosis. Scientific Reports 5, 16900-16900 (2015).
33. Zhang, S. & Qi, Q. MTSS1 suppresses cell migration and invasion by targeting CTTN in glioblastoma. Journal of Neuro-oncology 121, 425-431 (2015).
34. Wang, P. et al. PFDN1, an indicator for colorectal cancer prognosis, enhances tumor cell proliferation and motility through cytoskeletal reorganization. Medical Oncology 32, 264-264 (2015).
35. Gu, S. et al. Human Dental Pulp Stem Cells via the NF-κB Pathway. Cellular Physiology and Biochemistry 36, 1725-1734 (2015).
36. Huang, G. et al. Clinical and therapeutic significance of sirtuin-4 expression in colorectal cancer. Oncology Reports 35, 2801-2810 (2016).
37. Yan, X., Ye, T., Hu, X., Zhao, P. & Wang, X. 58-F, a flavanone from Ophiopogon japonicus, prevents hepatocyte death by decreasing lysosomal membrane permeability. Scientific Reports 6, 27875 (2016).
38. Ding, W., Tong, Y., Zhang, X., Pan, M. & Chen, S. Study of Arsenic Sulfide in Solid Tumor Cells Reveals Regulation of Nuclear Factors of Activated T-cells by PML and p53. Scientific Reports 6, 19793-19793 (2016).








