Antibody-drug conjugate (ADC) in clinical application (Approved/BLA, phaseI/II/III)
FDA approved Antibody-drug conjugate (ADC) for clinical use
Clinically, most of the ADC drugs approved by FDA are IgG1, and the targets are CD33, CD22, HER2 and so on. The most common payload is MMAE, also contains calicheamicin, DM1. ADC drugs are mainly used in the field of antitumor, which is one of the hot research directions in recent years. At present, 11 ADC drugs have been approved in the world, including Mylotarg (Pfizer), adcetris (Seattle genetics / Takeda), kadcyla (Roche), besponsa (Pfizer), lumoxiti (AstraZeneca), Polivy (Roche), padcev (Seattle genetics / anstelai / MSD), enhertu (AstraZeneca / first third party), trodelvy (immunomedicine), blenrep (GSK) Akalux (Rakuten Aspyrian)1. From the perspective of listed drug R & D enterprises, Pfizer, Seattle genetics, Roche and AstraZeneca have two models respectively, and the other three companies have one model respectively.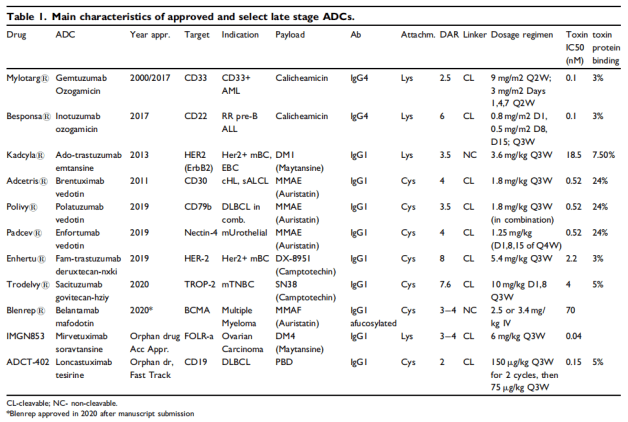
Antibody-drug conjugate (ADC) currently under clinical investigation
There are currently 82 novel ADCs in 150 active clinical trials registered with clinicaltrials.gov for cancer patients. Most of the ADCs are currently under investigation in phase 1 trials, while a small percentage has advanced to phase 3. Of the 150 ongoing trials, more than 80% are evaluating ADC safety and efficacy in solid tumors whereas less than 20% are trials for hematological malignancies. There are 43 disclosed targets organized here by the number of ADCs designed to recognize them. Most of these targets are under evaluation by a single ADC, while some are being investigated by several different ADCs. Of the 82 novel ADCs, followed by DNA-damaging molecules, topoisomerase I inhibitors, and finally unique payloads such as TLR agonists, a BCL2-xL inhibitor, and an RNA polymerase II inhibitor. Many payloads can’t be disclosed. Most ADCs under clinical investigation either utilize the conventional cysteine conjugation strategy or site-specific conjugation linker while few conjugate to surface lysines. 2Novel Antibody-drug conjugate (ADC) in Clinical Trials
More than 80 ADCs are currently in active clinical trials, with a majority in phase I and I/II. Over 80% of the clinical trials are investigating ADC safety and efficacy in various solid tumors, while the remaining trials involve hematological malignancies. HER2 is currently one of the most attractive targets for ADC development, with three anti-HER2 ADCs currently in phase III trials. One such anti-HER2 ADC is RC48, produced by RemeGen, joining an IgG1 anti-HER2 antibody, hertuzumab, to approximately four MMAE molecules via a protease-cleavable valine-citrulline linker through cysteine conjugation.3Reference:
1. Coats S, Williams M, Kebble B, Dixit R, Tseng L, Yao NS, Tice DA, Soria JC. Antibody-Drug Conjugates: Future Directions in Clinical and Translational Strategies to Improve the Therapeutic Index. Clin Cancer Res. 2019 Sep 15;25(18):5441-5448. doi: 10.1158/1078-0432.CCR-19-0272. Epub 2019 Apr 12. PMID: 30979742.
2. Abedi M , Cohan R A , Mahboudi F , et al. MALDI-MS: a Rapid and Reliable Method for Drug-to-Antibody Ratio Determination of Antibody-Drug Conjugates[J]. Iranian biomedical journal, 2019, 23(6).
3. Xuejing, Yao, Jing, et al. A novel humanized anti-HER2 antibody conjugated with MMAE exerts potent anti-tumor activity[J]. Breast Cancer Research&Treatment, 2015.






 View the Knowledge base of Antibody-drug Conjugate (ADC)
View the Knowledge base of Antibody-drug Conjugate (ADC)