Review for ADC production, quality control and functional assay
SDS-PAGE
We need run SDS-PAGE(reducing and non-reducing DTT) to see the integrity of antibody and Preliminary observation of the connection of small molecules. In general, we can see that the conjugated antibody will shift upward compared with the naked antibody
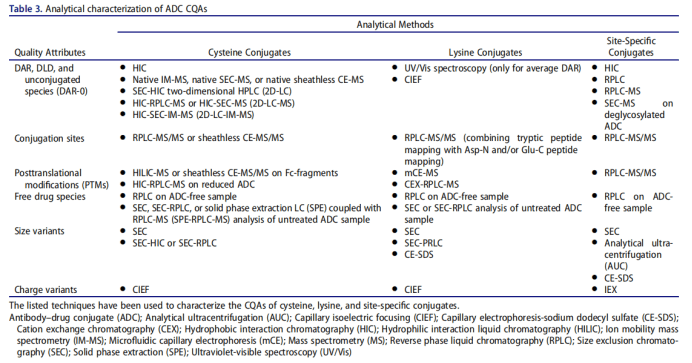
DAR (methods and standard)
ADC drugs are essentially a mixture, which is composed of mAbs connecting different numbers of small molecule drugs. Dar represents the average number of small molecule drugs connected to each mAb. Dar directly affects the efficacy and safety of ADC drugs. In the drug development stage, the variation range of DAR value should be minimized. The coupling sites of ADC drugs are divided into amino groups on lysine residues and sulfhydryl groups on cysteine residues. The DAR coupled by lysine is often small, but there are many potential coupling sites. The coupling reaction is random and the product heterogeneity is large; There are 4 pairs of inter chain disulfide bonds used in the development of ADC drugs. The antibody converts the inter chain disulfide bond into free cysteine residues through partial reduction. The sulfhydryl group in the cysteine residues reacts with the maleimide group in the linker to form ADC. Currently, most ADCs in use or under development rely on interchain disulfide cysteines for conjugation, in which the 4 (IgG1 and IgG4) or 6 (IgG2) interchain disulfide bonds are reduced by an excess reducing agent, namely tris(2-carboxyethyl)phosphine or dithiothreitol.153 This spares disruption of intrachain disulfide bonds while freeing sulfhydryl groups from cysteine residues participating in interchain disulfide bonds (Figure 3e). The resulting product is a mixture of ADCs containing 0–8 drugs per parent IgG1 or IgG4 and 0– 12 per IgG2, with predominantly even numbered DAR (0, 2, 4, 6, 8, 10, 12) species within the ADC mixture.
1. Ultraviolet / visible spectroscopy (UV / VIS)
UV / Vis spectroscopy is the simplest and stable method to detect Dar value. This method requires antibodies and small molecule drugs to have different maximum absorption wavelengths, and calculate their concentrations respectively to obtain the DAR value of ADC, which is suitable for a variety of ADCs.
2. Chromatography
Chromatography includes hydrophobic interaction chromatography (HIC) and reverse phase high performance liquid chromatography (RP-HPLC), which are suitable for the determination of cysteine coupled ADC. Hydrophobic interaction chromatography can separate the components with different Dar values according to the difference of hydrophobicity, and maintain the structural integrity of ADC molecules; Reversed phase high performance liquid chromatography needs to reduce the antibody to obtain light and heavy chains before analysis. It can be used to supplement and verify the results of hydrophobic interaction chromatography, and is suitable for mass spectrometry.
3. Mass spectrometry
Mass spectrometry is suitable for the determination of DAR value of lysine coupled ADC, including liquid chromatography tandem mass spectrometry and MALDI-TOF-MS. Lysine coupled ADC has strong heterogeneity, which increases the difficulty of mass spectrum analysis. Usually, additional pretreatment of ADC is required before determination, such as deglycosylation and removal of C-terminal lysine heterogeneity.
Cytotoxity assay of ADC
After the DAR of antibody is determined, the cytotoxicity experiment of ADC needs to be carried out. The cells are treated with different doses of ADC and their EC50 is analyzed. In the cytotoxicity experiment in vitro, the cells in the control group knocked out the target gene, so the antibody lost the role of localization and could not kill tumor cells, so it did not have the regularity of dose. However, from the treatment results of cells that have never been knocked out, they generally have better killing and EC50.






 View the Knowledge base of Antibody-drug Conjugate (ADC)
View the Knowledge base of Antibody-drug Conjugate (ADC)