Internal Gene Editing Field Analysis
| Cat No. | Products Name | Type of Crispr | Viral vector | Promoter | Fluorescent/Resistance | Tag | Order |
| GMV-Crispr-AAV001 | AAV-CMV-saCas9 | saCas9 | AAV | CMV | Null | HA |  |
| GMV-Crispr-AdV001 | Ad-CMV-spCas9 | spCas9 | Adenovirus | CMV | zsgreen | FLAG |  |
| GMV-Crispr-AdV002 | Adv-CMV-spCas9-zsgreen | spCas9 | Adenovirus | CMV | EGFP | FLAG |  |
| GMV-Crispr-AdV003 | Adv-CMV-spCas9-EGFP | spCas9 | Adenovirus | CMV | Null | FLAG |  |
| GMV-Crispr-AdV004 | Adv-CMV-spCas9-mcherry | spCas9 | Adenovirus | CMV | mcherry | FLAG |  |
| GMV-Crispr-LV001 | Lv-CMV-spCas9-puromycin | spCas9 | lentivirus | CMV | puromycin | FLAG |  |
| GMV-Crispr-LV002 | Lv-CMV-spCas9-zsgreen | spCas9 | lentivirus | CMV | zsgreen | FLAG |  |
| GMV-Crispr-LV003 | Lv-CMV-spCas9-EGFP | spCas9 | lentivirus | CMV | EGFP | FLAG |  |
| GMV-Crispr-LV004 | Lv-CMV-spCas9-mcherry | spCas9 | lentivirus | CMV | mcherry | FLAG |  |
| GMV-Crispr-LV005 | Lv-CBH-spCas9-puromycin | spCas9 | lentivirus | CBH | puromycin | FLAG |  |
| GMV-Crispr-LV006 | Lv-CBH-spCas9-zsgreen | spCas9 | lentivirus | CBH | zsgreen | FLAG |  |
| GMV-Crispr-LV007 | Lv-CBH-spCas9-EGFP | spCas9 | lentivirus | CBH | EGFP | FLAG |  |
| GMV-Crispr-LV008 | Lv-CBH-spCas9-mcherry | spCas9 | lentivirus | CBH | mcherry | FLAG |  |
Custom made CRISPR/Cas9 service
CRISPR/Cas9 User Manual
Crispr/cas9 mediated Gene knockout  |
AAV Production CRISPR/Cas9 Knockout System-User Manual  |
Adenovirus CRISPR/Cas9 Knockout System-User Manual  |
Lentivirus-CRISPR/Cas9 Knockout System-User Manual  |
Editas Medicines
Genome editing firm that researches and develops therapies for treating genetic based diseases primarily using CRISPR technology.
CEO: Cynthia Collins
Website: https://www.editasmedicine.com/
Located: Cambridge, Massachusetts
Employees: 133
Market Cap: $22.06 (use link for updated value)
Annual Revenue (2018): $30.1M
Funding History: Series A funding of $43M via Flagship Ventures achieved in November 2013, Series B funding of $120M via Deerfield achieved in August 2015, and IPO of $94.4M achieved in February 2016.
Investment: $87M Series A investment in Beam Therapeutics on May 13, 2018.
Pipeline: Information about Editas’ pipeline is shown here. Included in the link is information regarding Editas’ editing mechanisms, delivery modes, and commercial rights.
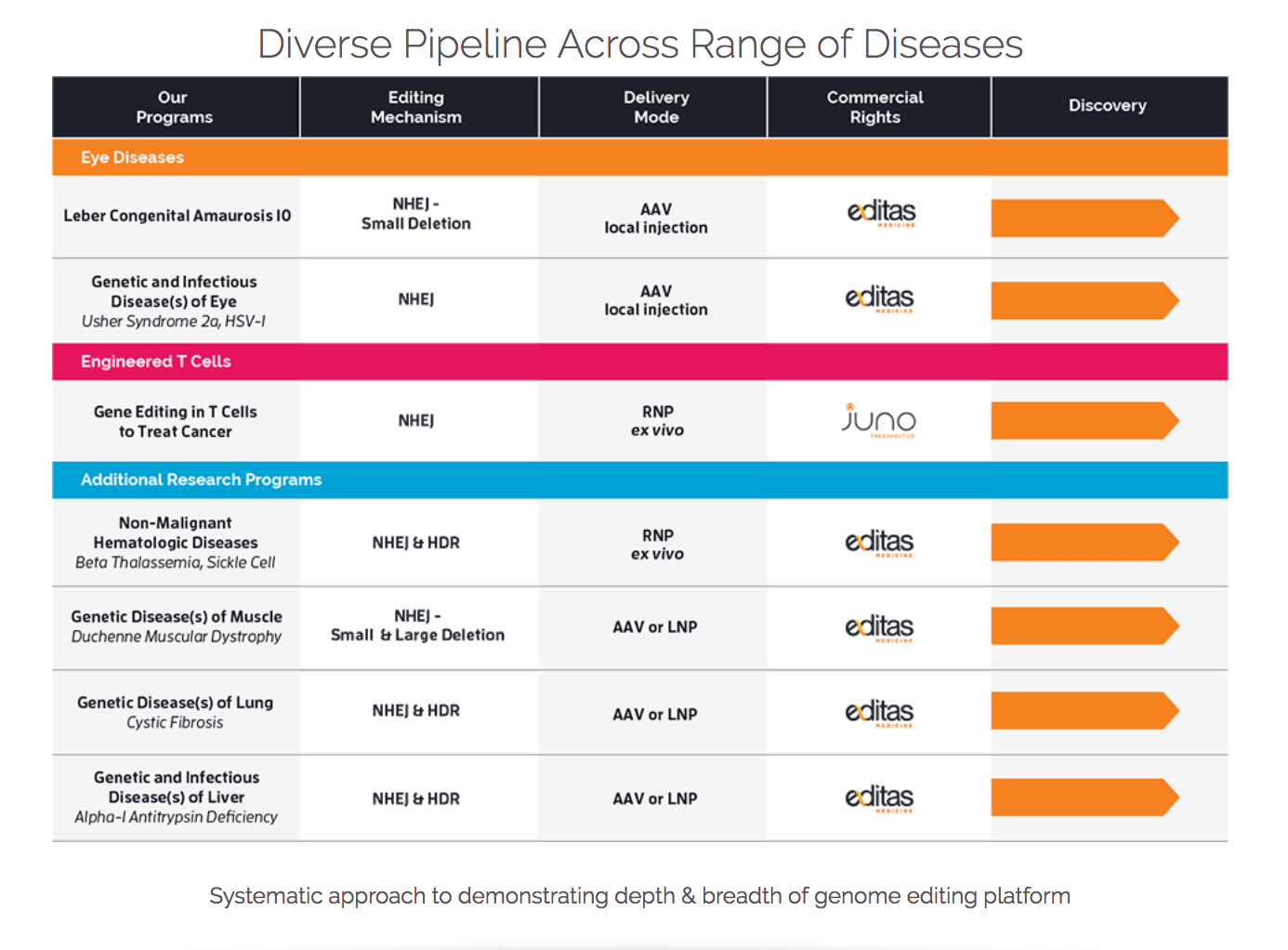
License History, Partnerships, & News
October 2019: Editas and AskBio (AAV company) enter into a strategic research collaboration to explore in vivo possibilities for treating neurological diseases.
July 2019: Brilliance clinical trial announced, the first study of CRISPR inside the body. It is a Phase 1/2 study to evaluate AGN-151587 for the treatment of LCA10, assessing safety, tolerability, and efficacy in approximately 18 patients.
May 30, 2019:
Editas Medicine has exclusively licensed a newly released CRISPR enzyme from Integrated DNA Technologies for human therapeutic applications, IDT said today. The new enzyme, Alt-R Cas12a (Cpf1) Ultra is a mutant of Acidaminococcus sp.BV3L6 Cas12a (Cpf1) that has enhanced editing activity, reaching or exceeding the performance of Cas9, according to IDT.
April 3, 2019 (BlueRock): Editas and BlueRock Therapeutics enter strategic research collaboration and cross-licensing agreement to combine genome editing and cell therapy platforms. Collaboration allows Editas to discover and develop engineered cell medicines across oncology, including solid tumors and blood cancers.
December 9, 2016: Exclusively licensed intellectual property related to new CRISPR technologies for human therapeutics that will enhance and broaden the range of medicines the company can develop. These global licensing agreements include intellectual property owned by the Broad Institute of MIT and Harvard (Broad Institute), Harvard University, Massachusetts Institute of Technology (MIT), Wageningen University, the University of Iowa, and the University of Tokyo for Cpf1 and advanced cases of Cas9.
August 3, 2016 (MGH): Entered into an exclusive license agreement with Massachusetts General Hospital to access intellectual property and technology related to high-fidelity Cas9 nucleases and Cas9 PAM variants that will enable the company to address an expanded range of genetically-defined diseases with the potential for enhanced specificity.
Dec 1, 2014 (Duke): Editas entered into an exclusive license agreement with Duke University to access intellectual property and technology related to the CRISPR/Cas9 and TALEN genome editing systems.
Dec 1, 2014 (MGH): Editas enters exclusive license agreement with Massachusetts General Hospital to access intellectual property and technology related to the CRISPR/Cas9 and TALENs genome editing systems. The license allows broad utilization of the technology developed in the laboratory of Editas founder, J. Keith Joung, M.D., Ph.D., for the prevention and treatment of human or animal disease, and broad agricultural use.
News
January 22, 2019: Katrine Bosley steps down as CEO, replaced by director Cynthia Collins to serve as interim CEO.
November 30, 2018: FDA accepts Editas’ IND application for an experimental CRISPR genome editing medicine for the treatment of Leber Congenital Amaurosis type 10 (LCA10).
Intellia Therapeutics
Uses genome editing methods such as CRISPR/Cas9 to treat diseases or correct genetic causes of disease.
CEO: John Leonard
Website: https://www.intelliatx.com/
Location: Cambridge, Massachusetts
Employees: 211
Market Cap: $14.97 (use link for updated value)
Annual Revenue: $30.4M (2018)
Funding History: $15M Series A funding in November 2014 via Atlas Venture. $70M Series B funding in August 2016 via OrbiMed. IPO in May 2016 of $108M.
Pipeline: Information regarding Intellia’s pipeline is detailed here.
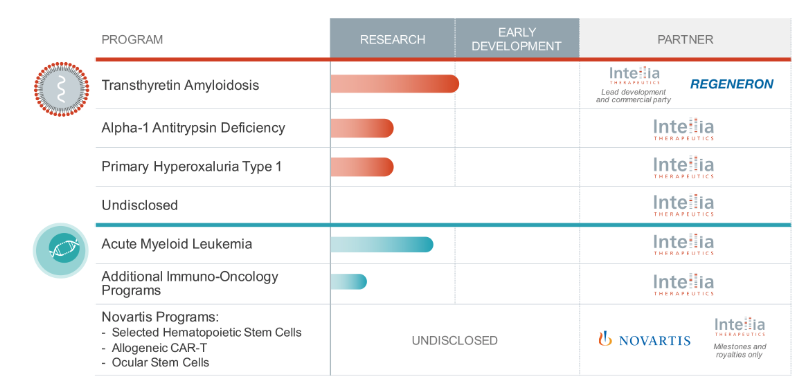
Partnerships & Licenses
Novartis (2014): Strategic partnership and licensing agreement that will accelerate the development of ex vivo CRISPR/Car9 using CARTS and HSCs.
Regeneron (2016): License and collaboration agreement centered around the research, development, and commercialization of CRISPR/Cas-based therapeutic products primarily focused on genome editing in the liver. Intellia and Regeneron will also perform research and development activities aimed at discovering and developing novel technologies and improvements to CRISPR/Cas technology to enhance the genome editing platform.
Caribou Biosciences (2014): Licensing and service agreement in which Caribou granted Intellia an exclusive license to its CRISPR/Cas9 intellectual property for human therapeutic use excluding anti-fungal and anti-microbial applications.
Ospedale San Rafaele (2017): Three-year research collaboration, option and license agreement to engineer optimized T cell-based cancer therapies. The goal of the collaboration is to discover innovative tools to target tough-to-treat cancers, while leveraging Intellia’s proprietary CRISPR/Cas9 platform to generate next-generation T cell therapies that will address unmet needs in both hematological and solid tumors.
News
May 28, 2019: Intellia names Laura Sepp-Lorenzino as its new Executive Vice President and Chief Scientific Officer.
April 2019: Intella presents new in vivo and engineered cell therapy data at the America Society of Gene and Cell Therapy, including the first demonstration of targeted gene insertion with CRISPR/Cas9 in the liver of non-human primates.
October 31, 2018: Intellia files for arbitration alleging Caribou broke their 2014 agreement. As of May 20, 2019, the arbitration is ongoing.
CRISPR Therapeutics
Uses CRIPSR/Cas9 in four capacities: hemoglobinopathies, in vivo, immuno-oncology, and regenerative medicine.
CEO: Samarth Kulkarni
Website: http://www.crisprtx.com/#programs
Location: Switzerland and Cambridge, Massachusetts
Employees: 188
Market Cap: $37.69 (click for updated value)
Annual Revenue: $2.1M
Funding History: Versant Ventures Series A investment in April 2015 of $35M, and a series B investment in the same month of $29M. Second round of series B by Franklin Templeton Investments yielded $38M. IPO in October 2016 yielding $56M, and investment of $35M in exchange for equity.
Pipeline: Information regarding CRISPR therapeutics’ pipeline is located here.

Partnerships & Licenses
October 2019: Entered licensing agreement with KSQ Therapeutics, a company that uses a CRISPR platform to gauge the probability of drug success.
Vertex: CRISPR Therapeutics established a four-year strategic research collaboration with Vertex Pharmaceuticals. The initial research focuses on discovering treatments to address the underlying genetic causes of cystic fibrosis and hemoglobinopathies, specifically sickle cell disease and β-thalassemia. The collaboration was extended for two more years in June 2019.
Casebia: CRISPR Therapeutics and Bayer formed Casebia Therapeutics to bring therapies to patients suffering from blood disorders, blindness and congenital heart disease. CRISPR brings CRISPR/Cas9 gene-editing technology to Casebia. CRISPR has 50 percent ownership in Casebia, which is fueled by $300 million in research and development funding from Bayer over five years.
ViaCyte: CRISPR Therapeutics established a five-year collaboration with ViaCyte, Inc. focused on the discovery, development, and commercialization of gene-edited allogeneic stem cell therapies for the treatment of diabetes. Upon successful completion of these studies and identification of a product candidate, the parties will jointly assume responsibility for further development and commercialization worldwide.
January 22, 2019 (ProBioGen): CRISPR Therapeutics and ProBioGen sign collaboration and license with agreement to develop novel in vivo delivery technologies.
November 9, 2018 (MaxCyte): CRISPR Therapeutics and MaxCyte Expand Clinical and Commercial License Agreement into Oncology.
March 28, 2018 (Patent): CRISPR Therapeutics Announces European Patent for CRISPR/Cas Gene Editing.
March 14, 2017 (MaxCyte): CRISPR Therapeutics and Casebia Therapeutics announce Commercial License Agreement with MaxCyte. CRISPR Therapeutics and Casebia obtain commercial rights to MaxCyte’s cell engineering platform to develop CRISPR/Cas9-based therapies.
Caribou Biosciences
Caribou Biosciences is a private company that develops genome-based technology solutions for cellular engineering and analysis.
CEO: Rachel Haurwitz
Website: https://cariboubio.com/
Location: Berkley, California
Employees: 20
Funding: $11M Series A investment from Novartis in April 2015, $30M Series B investment from seven investors in May 2016.
Annual Revenue: Estimated $4M
Licenses & Partnerships
Exclusive license to CRISPR-Cas9 from the University of California and the University of Vienna.
Oxford Nanopore Technologies (2019): Caribou and company ONT strike licensing agreement for advancement of their nanopore sequencing technology.
RenoVAte Biosciences (2019): Non-exclusive license agreement in which RenoVAte can use CRISPR-Cas9 for research purposes on cattle, sheep, and pigs.
Medical Research Council (2019): MRC has a non-exclusive, worldwide license to create genetically engineered mice for research purposes.
Genus plc (2016): Multi-year strategic research collaboration in which Caribou provides Genus with exclusive access to Cas9 for the development of new traits in pigs and cattle.
Integrated DNA Technologies, Inc. (2016): Non-exclusive license agreement in which Caribou grants IDT worldwide rights to commercialize CRISPR and Cas9 reagents.
The Jackson Laboratory (2016): JAX has non-exclusive, worldwide rights to use Caribou’s CRISPR-Cas9 IP to create genetically engineered mice for research purposes.
Novartis (2014 & 2016): Caribou granted an option for a non-exclusive, worldwide license for internal research using CRISPR and Cas9 for drug target screening and validation techniques, this license was extended in 2016.
DuPont Pioneer (2015): License agreement and multi-year collaboration that includes cross-licensing of key IP for both companies (CRISPR and Cas9).
Intellia Therapeutics (2014): Caribou a co-founder of Intellia, and Intellia has exclusive access to Caribou’s CRIPSR and Cas9 technology to develop gene, cell, and anti-viral therapies. In addition, Caribou has access to IP developed by Intellia that is outside their field of operation.
News
Caribou and Intellia are involved in an arbitration battle regarding the use of Intellia’s technology.
AstraZeneca
AstraZeneca develops pharmaceutical solutions for cancer, cardiovascular, gastrointestinal, infection, neuroscience, respiratory and inflammation.
CEO: Pascal Soriot
Website: https://www.astrazeneca.com/
Location: Cambridge, United Kingdom
Employees: 61.1K
Market Cap: $37.81 (click link for updated value)
Annual Revenue: $21.9B
Acquisitions: Acerta Pharma (oncology) for $4B, ZS Pharma for $2.7B.
Pipeline: AstraZeneca has a diverse pipeline. Information involving CRISPR is shown in the oncology section.
CRISPR Collaboration:
Wellcome Trust Sanger Institute (research)
Innovative Genomics Initiative (research)
Thermo Fisher Scientific (screening)
Broad Institute/Whitehead Institute (goal to identity new targets for cancer drug discovery.
Beam Therapeutics
Beam therapeutics is a private company that develops precision medicines through base editing technology to treat genetic diseases.
CEO: John Evans
Website: https://beamtx.com/
Location: Cambridge, Massachusetts
Estimated Employees: 70
Estimated Annual Revenue: $1M
Funding History: $87M Series A investment in 2018, $135M Series B in 2019.
Pipeline: Gene correction, gene regulation, gene slicing, and gene programming. More information can be found here.
Licenses & Partnerships
May 30, 2019: Beam Therapeutics and Bio Palette (Japanese base-editing company) announced an exclusive cross-license agreement around each companies’ base editing intellectual property.
Harvard: License agreement with Harvard for two base editing platforms.
Editas: Licensing and option agreement for exclusive rights to certain IP licensed to Editas by Harvard, the Broad Institute, and MGH.
Casebia Therapeutics
Co-founded by CRISPR Therapeutics and Bayer, CASEBIA discovers, develops and commercializes therapeutics to cure blood disorders, blindness and congenital heart disease.
CEO: James Burns
Location: Cambridge, Massachusetts
Estimated Number of Employees: 80
Estimated Annual Revenue: $10.2M
Funding History: $335M investment from Bayer.
Pipeline: Autoimmunity, retinal and liver programs.
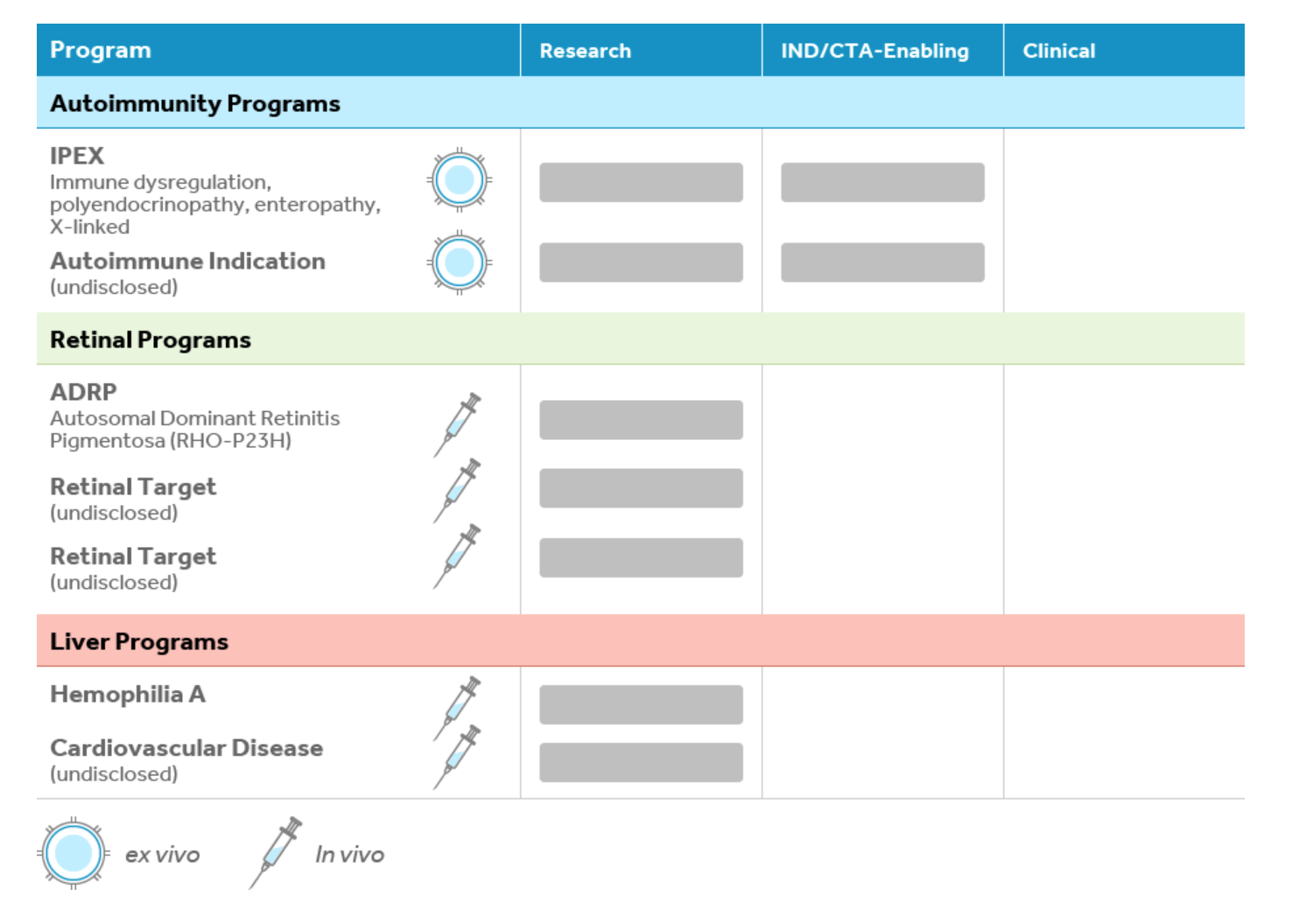
License History & Partnerships
Seattle Children’s Research Institute (2017): Exclusive license agreement and research collaboration to prevent autoimmune diseases using CRISPR/Cas9.
StrideBio (2017): Casebia gets to use StrideBio’s AAV vectors for in vivo CRISPR/Cas9 based therapies in exchange for funding, milestones and royalties on licensed vectors, and retention to use novel AAV vectors for gene therapy applications.
CureVac (2017): Collaboration between CRISPR Therapeutics, Casebia, and CureVac in which CureVac will develop novel Cas9 mRNA constructs with improved properties for gene editing applications, such as increased potency, decreased duration of expression, and reduced potential for immunogenicity. CureVac will receive an upfront payment, research funding, and will be eligible to receive development and commercial milestones and royalties on commercialized products arising from the collaboration.
News
June 5, 2019: Casebia announces it has appointed Stephen Kennedy as Senior Vice President and Head of Technical Operations.
Refuge Biotech
Refuge Biotech is a private company that develops a genetic engineering platform focusing on gene editing of immune cells for cancer immunotherapy using “advanced” CRISPR methods. More information on their methodology shown here.
Website: https://refugebiotech.com/
Location: Menlo Park, California
Estimated Employees: 30
Estimated Annual Revenue: $3M
Funding History: Series A funding of $8.3M and Series B funding of $25M
Pipeline:
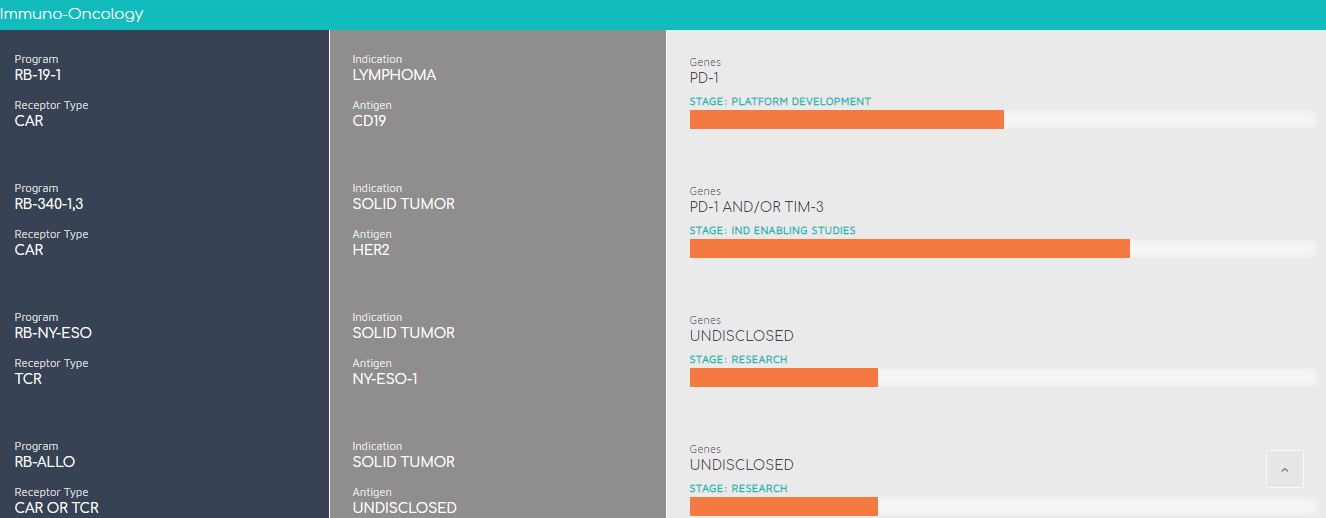
Licenses & News:
April 2018: Research collaboration with Chinese pharmaceutical company “3sBio”, focusing on developing Programmed Therapeutic Cells that can produce therapeutic biologics agents in a disease micro environment inside patient’s body, using Refuge’s platform technology. 3SBio will have exclusive license to develop and commercialize the Programmed Therapeutic Cells in Greater China under the research collaboration agreement.
Inscripta
Inscripta is a private company that develops and commercializes CRISPR enzymes for gene-editing research solutions.
Website: https://www.inscripta.com/
Location: Boulder, Colorado
Employees: 45
Funding History: $6M Series A funding, $23M Series B funding in 2017, $105.5M Series C funding in 2019.
News
September 2018: Inscripta acquires Solana. Through the acquisition, Inscripta and Solana have joined forces to accelerate the commercialization of Inscripta’s suite of gene-editing technology tools. Inscripta’s acquisition of Solana assembles an elite, life-sciences product development and manufacturing pipeline for precision gene editing.
July 2018: Inscripta is granted a patent for its “Mad7” CRISPR enzyme.
December 2017: Inscripta releases Mad7, a CRISPR enzyme, for free to all researchers, and at a single digit royalty price for all non-R&D purposes.
Exonics Therapeutics
Exonics is a private biotechnology company that focuses on providing gene repair therapies for genetic neuromuscular diseases, specifically duchenne muscular dystrophy.
Website: http://exonicstx.com/
Location: Watertown, Massachusetts
Employees: 46
Estimated Annual Revenue: $9.2M
Funding History: $40M Series A funding and $5M seed/angel funding in 2017.
News
June 2019: Vertex announced plans to acquire Exonics Therapeutics for approximately $1 billion.
Mammoth Biosciences
Co-founded by Jennifer Doudna, Mammoth Biosciences develops a CRISPR-based platform for detecting diseases and illnesses.
Website: https://mammoth.bio/
Location: San Francisco, California
Employees: 20
Funding History: $23M in funding over four rounds.
News:
Sherlock Biosciences
CRISPR based disease detecting technology. Co-founded by Feng Zhang.
Website: https://sherlock.bio/
Location: Cambridge, Massachusetts
Estimated Employees: 15
Funding History: $35M in financing.
News:
Precision Biosciences
Precision develops a genome editing platform that provides immuno-oncology therapeutics for the treatment of cancer.
CEO: Matthew Kane
Website: https://precisionbiosciences.com/
Location: Durham, North Carolina
Employees: 127
Market Cap: $13.50 (click for updated number)
Annual Revenue: $21.8M
Funding History: Series A $25.6M via VenBio in April 2015, Series B of $88.M via Amgen in May 2018, $39.5 debt investment in February 2019, and IPO of $145.4M in March 2019.
Pipeline: Target areas are Cancer immunotherapy, gene therapy, food, and genome editing, but nothing is currently in the pipeline.
Licenses & Partnerships
MaxCyte (2018): Clinical and commercial license agreement revolving around the use of ARCUS to treat a broad range of cancers.
University of Pennsylvania (2018): Collaboration to pursue research and development of gene editing therapies. This relationship will allow Precision and Penn to design new in vivo gene therapies based on Precision’s proprietary ARCUS genome editing technology.
Gilead Sciences (2018): Entered into a strategic collaboration to eliminate HBV using ARCUS.
Baxalta (2016): global collaboration to develop a broad series of allogeneic CAR T cell therapies directed towards areas of major unmet need in multiple cancers.
News:
July 2019: Precision announces opening of manufacturing facility in Durham, NC for their CAR-T cell therapies.
Sangamo Therapeutics
Sangamo focuses on the research, development and commercialization of deoxyribonucleic acid binding proteins for therapeutic gene regulation, using ZFN.
CEO: Sandy Macrae
Website: https://www.sangamo.com/
Location: Richmond, California
Employees: 302
Market Cap: $10.08 (click for updated value)
Annual Revenue: $79.9M
Acquisitions: Sangamo acquired TxCell S.A. in July 2018 for $84.4M. TxCell S.A. develops personalized T cell immunotherapies for the treatment of inflammatory and autoimmune diseases.
Funding History: IPO $52M in 2000.
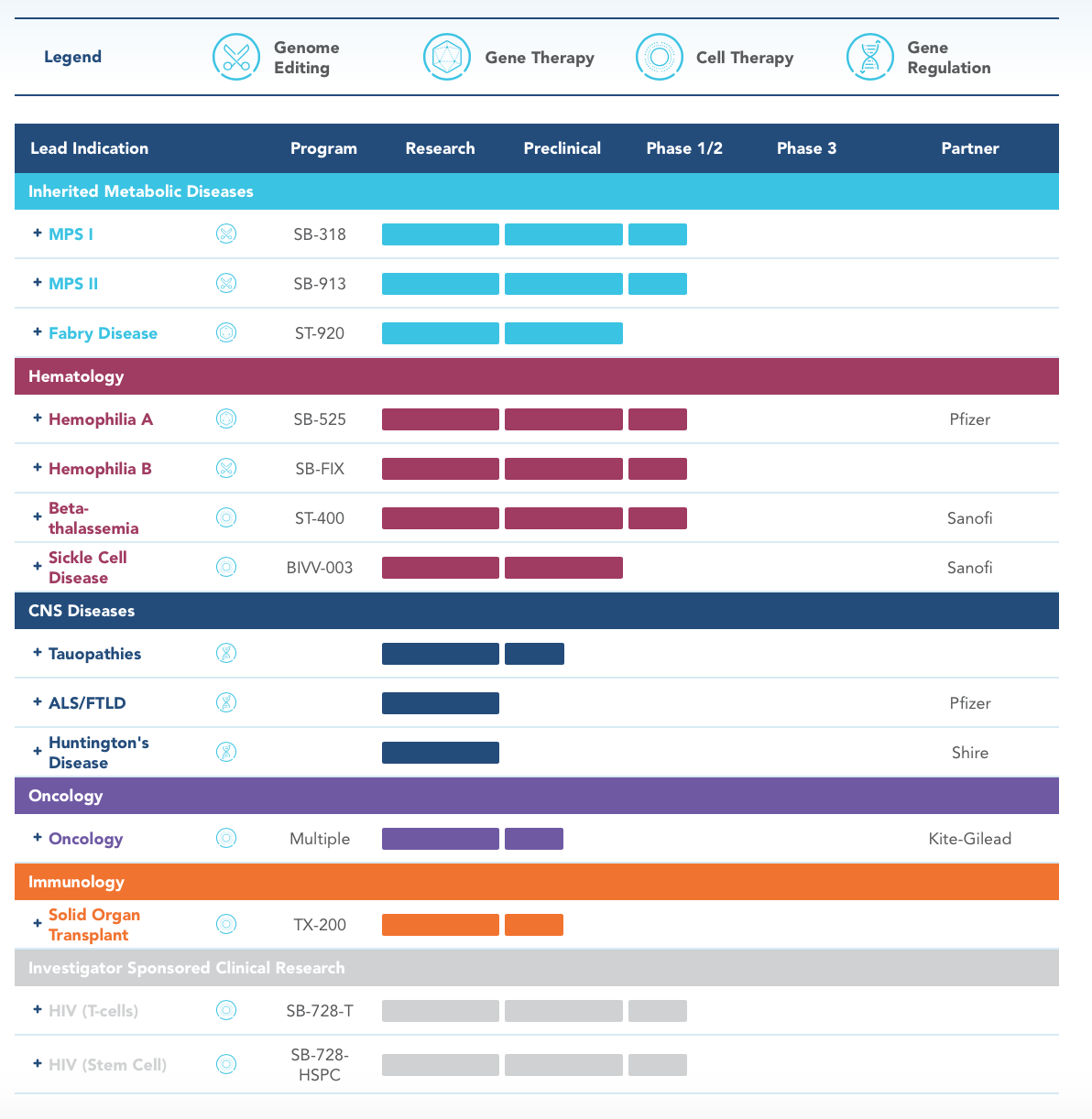
Licensing History & Partnerships
Kite: Kite, a Gilead Company, and Sangamo are developing next-generation ex vivo cell therapies to treat cancer by using Sangamo’s zinc finger nuclease (ZFN) technology.
Pfizer: Pfizer and Sangamo have an exclusive, global collaboration and license agreement to develop and commercialize gene therapies for hemophilia and ALS.
Sanofi: Sanofi and Sangamo are developing gene-edited cell therapies for the treatment of beta thalassemia and sickle cell disease, using Sangamo’s zinc finger nuclease technology.
Shire International GmbH: Shire plc and Sangamo are developing therapeutics for Huntington’s disease utilizing our ZFP-TF gene regulation platform.
Gilead Sciences: To work on ZFN approaches that may improve cancer treatment. As part of that deal, Sangamo received an upfront payment of $150 million from Gilead Sciences and it could receive up to $3.01 billion in potential milestones. If any therapies from this collaboration win approval, then Sangamo will receive tiered royalties on future sales.
News
May 14, 2019, Sangamo’s finance chief stepped down, effective in June 2019.
ORIG3N
ORIG3N is a private company that develops treatments for rare genetically inherited diseases with a focus on heart, liver and neurodegenerative indications. They gain access to cells for research through DNA tests sold to consumers both online and through sponsorship at sporting and fitness events.
Website: https://www.orig3n.com/
Location: Boston, Massachusetts
Estimated Employees: 60
Estimated Annual Revenue: $3M
Funding History: $14.4M Series B investment in July 2018, and $50M raised since 2015. Their primary investment came from Haitong International, an investment bank based in Hong Kong.
Offerings: Product offerings can be found here.
Partnerships & News:
January 2019: U.S. Ski & Snowboard, the Olympic National Governing Body (NGB) of ski and snowboard sports in the USA, and Orig3n, Inc., the leader in lifestyle genetic testing, are excited to announce Orig3n as the Official Genetics Partner of the U.S. Ski & Snowboard Team. Orig3n will be able to set up booths at events to collect cells.
November 2017: Acquisition of Interleukin Genetics CLIA laboratory and their remaining assets. Interleukin Genetics’ technology will become a core component of Orig3n’s DNA test business to empower more people to take charge of their health through genetic insights.
March 2016: ORIG3N, Sharp Edge Labs launch drug discovery collaboration in which ORIG3N will make patient-derived cells available to Sharp Edge Labs for use in its drug discovery platform for genetic disorders.
Applied StemCell
ASC engages in developing and commercializing solutions and tools for stem cell research applications. They offer various products and services which are shown in the previous links.
Website: https://www.appliedstemcell.com/
Location: Milpitas, California
Estimated Employees: 49
Estimated Annual Revenue: $11.4M
Funding History
July 2018: Applied StemCell secured $44 Million in its second institutional financing round, funded primarily through investment firms from China & Hong Kong.
Licenses, Partnerships, and News:
May 2019: ASC Therapeutics, A Division of Applied StemCell, Inc. (ASC), announced today that it has obtained an exclusive therapeutics license from Expression Therapeutics, LLC (ET) for coagulation factor VIII expression technologies including a highly compact, yet potent, liver-directed promoter and matching liver-codon optimized factor VIII transgene for adeno-associated viral vector gene therapy of hemophilia A.
April 2019: Applied StemCell cures Hemophilia A in mice.
REGENXBIO
Website: https://www.regenxbio.com/Location: Rockville, MarylandEmployees: 192 Market Cap: Here.Annual Revenue: $87MFunding History: $7.9M equity in October 2013, $30M Series C in January 2015, $70.5M Series D funding, and IPO $138.6M in September 2015.License History and NewsMay 30, 2019: REGENXBIO announces completion of dosing for Phase I/IIa Clinical Trial of RGX-314 in Wet AMD.May 24, 2019: REGENXBIO gets first FDA approval of a gene therapy based on its proprietary NAV technology platform.
Spark Therapeutics
Spark Therapeutics Spark Therapeutics develops gene therapies for the treatment of hemophilia and neurodegenerative diseases using AAV technology.Website: http://sparktx.com/Location: Philadelphia, PennsylvaniaEmployees: 368Market Cap: Here.Annual Revenue: $35.3MFunding History: $50M Series A funding in October 2013, $73M Series B funding in May 2014, IPO $161M in January 2015, and a secondary offering of $94M in December of 2015Pipeline: Shown here.License History & Other News:January 2018: Spark Therapeutics Enters into a Licensing and Supply Agreement for Investigational Voretigene Neparvovec Outside the U.S.September 2017: Licensing agreement with Genethon, a non-profit research and development organization dedicated to the development of gene therapies for orphan genetic diseases from research to clinical validation, for the development and commercialization of an adeno-associated viral (AAV) gene therapy targeting the liver to address a rare genetic disease.
Sarepta Therapeutics Website: https://www.sarepta.com/ Location: Cambridge, Massachusetts Employees: 499 Market Cap: Here Annual Revenue: $323.4M Pipeline: May 8, 2019: Sarepta announces agreement with Nationwide Children’s Hospital for rights to its Gene Therapy Program to treat Limb-Girdle Muscular Dystrophy Type 2A, the most common form of Limb-Girdle Muscular Dystrophy. October 15, 2018: Sarepta and Lysogene announce exclusive license agreement for LYS-SAF302, a late-stage gene therapy for the treatment of MPS IIIA, and grant of option rights to an additional CNS gene therapy candidate.
Sarepta develops and delivers ribonucleic acid targeted therapeutics for the treatment of neuromuscular and infectious diseases, specifically DMD.

Licenses & Partnerships
Voyager Therapeutics
Gene therapy firm that develops treatment for fatal and debilitating diseases of the central nervous system.
Website: https://www.voyagertherapeutics.com/
Location: Cambridge, Massachusetts
Employees: 123
Market Cap: Here
Annual Revenue: $11.9M
Pipeline:
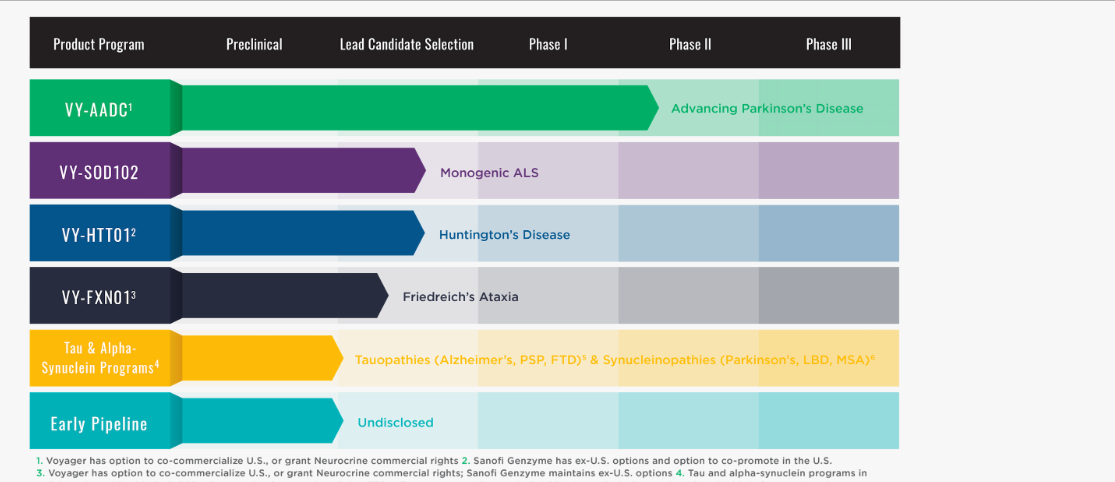
Licenses & Partnerships
February 22, 2019: Voyager Therapeutics and AbbVie announce collaboration to develop vectorized antibodies to treat Parkinson’s Disease and other synucleinopathies.
June 2014: Entered into a license agreement for use of REGENX’s proprietary NAV® vectors for the development and commercialization of gene therapies to treat Amyotrophic Lateral Sclerosis (ALS), Friedreich’s ataxia (FA) and Huntington’s disease (HD).
Poseida Therapeutics
Poseida Therapeutics is a private company that develops and commercializes gene therapies for the treatment of hematological malignancies and solid tumors.
Website: https://poseida.com/
Location: San Diego, California
Employees: 50
Estimated Annual Revenue: $3M
Pipeline:

Licenses & Partnerships
August 2018: Research collaboration and licensing agreement with Tenebio to develop novel CAR-T therapies using Teneobio’s heavy chain only domain antibodies (UniDabs™). Poseida will apply UniDab binders, which demonstrate significant advantages over traditional single chain variable antibody fragment (scFv) binders, to the development of its next generation CAR-T therapies.
August 2015: Worldwide License Agreement With Janssen to apply centyrin technology in the development of CAR Therapies
Vivet Therapeutics
Vivet Therapeutics is a private biotech company that develops and commercializes gene therapies for the treatment of liver disorders and orphan diseases.
Website: https://www.vivet-therapeutics.com
Location: Paris, France
Estimated Employees: 18
Pipeline:

Licenses & News
March 2019: Pfizer secures exclusive option to acquire Vivet. The companies are currently collaborating on the development of VTX-801, Vivet’s proprietary treatment for Wilson disease.
CIMA:
Fundacion Para La Investigacion Medica Aplicada (FIMA), a not-for-profit foundation part of the Center for Applied Medical Research (CIMA) of the University of Navarra and Vivet Therapeutics have formed a close collaboration involving the exclusive license to Vivet Therapeutics of FIMA’s novel proprietary AAV vector gene therapy technology to treat liver diseases such as Wilson disease.
Bluebird Bio
Bluebird focuses on developing transformative gene therapies for severe genetic diseases and cancer.
Website: https://www.bluebirdbio.com/
Location: Cambridge, Massachusetts
Employees: 764
Market Cap: Here
Annual Revenue: $51.1M
Pipeline: Better shown here than in picture format.
Licenses & News
July 2019: $6.12M investment from Marshall Wace LLP.
June 2019: European regulators signed off on gene therapy for beta-thalassemia. More information can be found here.
January 2019: Entered into an exclusive license agreement to research, develop and commercialize chimeric antigen receptor (CAR) T cell therapies using Inhibrx’s proprietary single domain antibody (sdAb) platform to multiple cancer targets.
August 2018: Entered into a strategic research and development collaboration with Gritstone Oncology to identify tumor-specific targets and natural T-cell receptors (TCRs) directed to those targets for use in our established cell therapy platforms.
May 2017: Established partnership with Novartis around its proprietary lentiviral vector platform.
Orchard Therapeutics
Orchard develops and commercializes transformative gene therapies for the treatment of life-threatening and orphan diseases.
Website: https://www.orchard-tx.com/
Location: London, England
Employees: 175
Market Cap: Here
Annual Revenue: $1.2M
Funding History
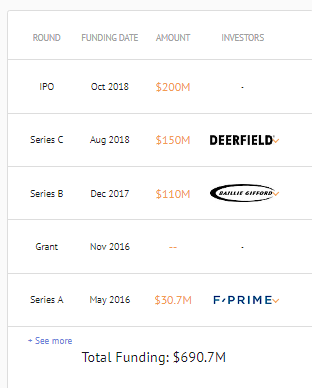
Pipeline: Shown here.
Licenses & News
May 2019: Exclusive global license with Fondazione Telethon and Ospedale San Raffaele in Milan, Italy for their ex vivo autologous hematopoietic stem cell (HSC) gene therapy program—a treatment for Mucopolysaccharidosis Type I (MPS-I) that has shown promising early clinical data in an ongoing Phase I/II proof of concept study.
December 2018: SIRION Biotech GmbH announced the entry into a license agreement, pursuant to which Orchard has licensed SIRION’s LentiBOOST™ technology to enhance manufacturing efficiency for certain of Orchard’s ex vivo autologous hematopoietic stem cell gene therapy drug candidates.
uniQure
uniQure is engaged in the development and commercialization of gene therapies for the treatment of cardiovascular and hemophilia diseases.
Website: http://uniqure.com/
Location: Amsterdam, Holland
Employees: 251
Market Cap: Here.
Annual Revenue: $8.9M
Pipeline:
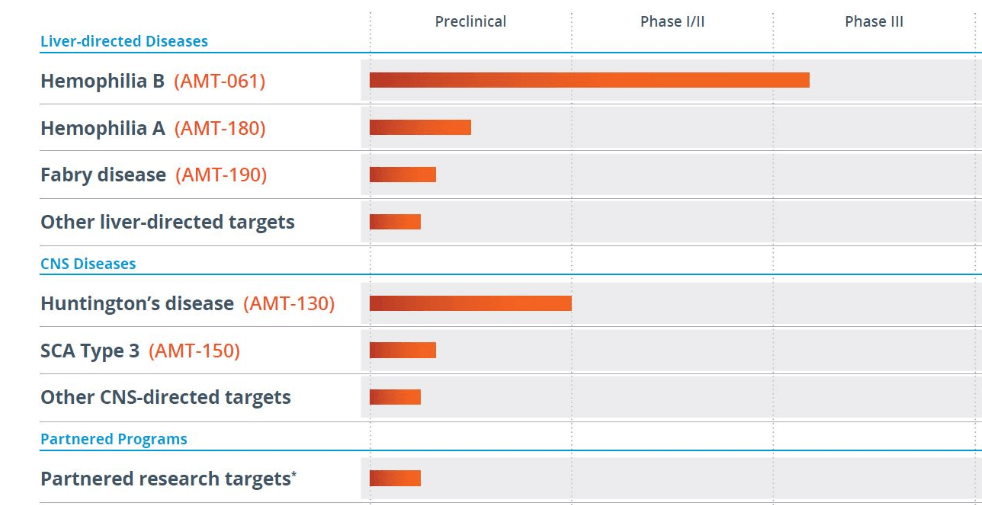
Partnerships & Licenses
Bristol-Myers Squibb: Agreement established in April 2015 that provides Bristol-Myers Squibb with exclusive access to uniQure’s gene therapy technology platform for multiple targets in cardiovascular diseases as well as the potential for target-exclusive collaboration in other disease areas. In total, the companies may collaborate on ten targets, including S100A1 for Congestive Heart Failure.
Molecular Therapeutics: uniQure has a collaboration and license agreement with 4D Molecular Therapeutics, signed in January 2014, focused on the identification of novel optimized AAV variants that have greatly enhanced properties for achieving higher levels of gene expression. The collaboration will evolve AAV variants with greatly improved gene delivery properties in liver and the central nervous system.
Sympronics: Entered into a co-operation and license agreement in January 2015 with Synpromics Limited for the discovery of alternative small liver-specific promoters for sustainable and increased expression of larger therapeutic genes fitting the package capacity of AAV vectors.
Illumina
Illumina develops, manufactures and markets integrated systems for the analysis of genetic variation and biological function.
Website: https://www.illumina.com/
Location: San Diego, California
Employees: 7,300
Market Cap: Here
Annual Revenue: $3.4B
Acquisition History (PacificBio acquired for $1.2B in November 2018)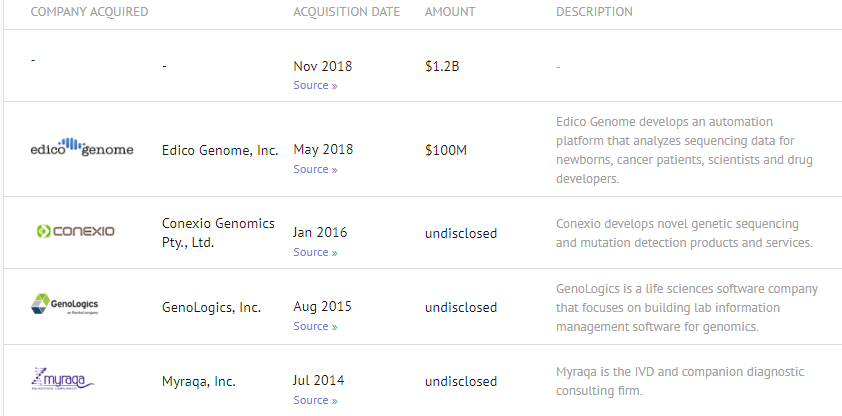
News
July 2019: Illumina, Inc announced that it has filed three patent infringement suits relating to BGI’s sequencing products, including the BGISeq-500, MGISeq-2000, and related chemistry reagents. The patents cover Illumina’s proprietary sequencing-by-synthesis chemistry. These lawsuits follow related patent suits filed by Illumina against BGI in Germany and Denmark.
June 2019: Illumina introduces expanded version of VeriSeq™ NIPT Solution, offering more comprehensive detection of rare chromosomal conditions.
May 2019: Illumina files patent infringement suit against BGI in Denmark.
January 2018: Thermo Fisher and Illumina announced they have signed a commercial agreement that enables Illumina to sell Ion AmpliSeq technology to researchers who conduct scientific studies on Illumina’s next-generation sequencing (NGS) platform.
MetaSystems
MetaSystems is a private company that develops and manufactures systems for automated microscope based imaging.
Website: https://metasystems-international.com/us/about/
Location: Germany and Newton, Massachusetts.
Estimated Employees: 132
Estimated Annual Revenue: $40M
Partnerships
Coplan: Established in May 2014, Coplan (Italy), a manufacturer of the automated microbiology specimen processor WaspLAB™, and MetaSystems announced that they have signed a global and exclusive agreement for the integration of the automatic microscopy slide scanning system Metafer with WASPLab™.
News
April 2019: MetaSystems presents machine learning implementation on scanning platform metafer for pre-classification of gram stain slides. More information can be found here.
January 2019: MetaSystems opens in an office in Buenos Aires, Argentina. The foundation is in response to the growing demand for technical knowledge in Latin America.
Cytocell
Cytocell, a private company, is a manufacturer of fluorescence in situ hybridizationy bridisation (FISH) probes that provides DNA screening solutions.
Website: https://www.cytocell.com/
Location: Cambridge, England
Employees: 88
Estimated Annual Revenue: $17.8M
News
February 2019: New Cytocell facility opens in Cambridge, UK.
2014: Acquired by Oxford Gene Technology (OGT).
Synthego
Synthego, a private company, is a provider of genome engineering solutions for CRISPR editing and research.
Website: https://www.synthego.com/
Location: Redwood City, California
Employees: 90
Estimated Annual Revenue: $13M
Funding History: Raised $8.3M in Series A in 2016, raised $41M in Series B in 2017, and raised $110M in Series C in 2018.
Partnerships
Thermo Fisher: In 2017, Synthego announced a new alignment with Thermo Fisher Scientific to manufacture and distribute synthetic guide RNA products. Synthego’s synthetic guide RNA portfolio and synthetic guide RNA manufacturing capabilities will be utilized to produce synthetic guide RNAs under Thermo Fisher’s Invitrogen TrueGuide brand that will be marketed and sold across Thermo Fisher channels worldwide.
News
June 2019: Synthego introduces the next generation of CRISPR knockout technology with the gene knockout Kit v2.
March 2018: Synthego Appoints Dr. Jennifer Doudna to advisory board to accelerate company’s genome engineering mission. Doudna to advise Synthego as the company develops technology and services to further the adoption of CRISPR, bringing access, precision and automation to genome engineering.
January 2018: Synthego announces the launch of ICE (Inference CRISPR Edits), a new tool named. ICE software offers rapid, reproducible analysis of Sanger data, which costs a hundred times less than next-gen sequencing (NGS). ICE is free to use and open source, and the only CRISPR analysis software featuring batch processing of Sanger data.
Horizon Discovery
Horizon Discovery provides cell panel screening, data analysis, model generation, apoptosis assays and custom breeding solutions for drug manufacturers.
Website: https://www.horizondiscovery.com/
Location: Cambridge, England
Employees: 500
Estimated Annual Revenue: $52.5M
Funding History: Total of $57.1M raised over seven rounds.
Licenses & Partnerships
List of partnerships shown here.
May 2017: Pre-existing non-exclusive, worldwide license agreement with ERS Genomics Ltd. to significantly expand Horizon’s license coverage for the use of the CRISPR gene editing technology. This will enable Horizon to use CRISPR in multiple new areas across its products and services.
Thermo Fisher Scientific
Thermo Fisher is a life science research institution focused on providing laboratory equipment, chemicals, supplies and services.
Website: https://www.thermofisher.com/us/en/home.html
Location: Waltham, Massachusetts
Employees: 70k
Market Cap: Here
Annual Revenue: $24.6B
Pipeline: Information regarding Thermo’s gene editing products are here. product offerings, drug development, drug discovery, and service offerings are shown here.
Bionano Genomics
BioNano Genomics develops and markets genome mapping platform that enables researchers to identify and analyze structural variants in chromosomes.
Website: https://bionanogenomics.com/
Location: San Diego, California
Employees: 77
Market Cap: Here
Annual Revenue: $12.1M
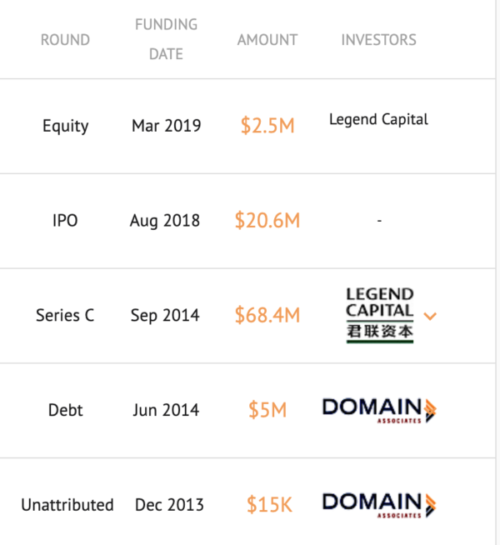
Funding History:
Partnerships & Licenses
October 2018: Bionano and Genoox launch integrated platform for identification of structural variants in DNA.
News:
February 2019: Bionano genomics’ Saphyr® system used for highly accurate detection of the genetic disorder FSHD as an alternative to southern blot.
Mission Bio
Mission Bio provides tools for the study of cellular heterogeneity in human health and life science research.
Website: https://missionbio.com/
Location: San Francisco, California
Employees: 75
Annual Revenue: $5M
Funding History: $10M Series A in 2017 and $30M Series B in 2018.
News:
May 2019: Published study using single-cell genomics to reveal clonal diversity underlying leukemia therapy resistance.
March 2019: Mission Bio launches Tapestri Designer, the first automated cloud tool for targeted single-cell DNA custom panels.




