Protocol for cell sorting
1. Remove spleens, LN, etc. into media on ice. Disrupt into single cell suspension using your favorite technique and pass through 70uM filter.
2. Centrifuge at 1500 RPM for 10 minutes at 8˚C.
3. Perform red blood cell lysis. Re-suspend in FACS staining buffer. (Use this buffer also for all washes until directed to use Sorting Buffer.) Adjust cells to 20-50 * 106/ml for typical staining reactions.
4. Add the appropriate number of cells to be stained into a FACS tube or 15mL conical tubes.
5. Block with Fc Block (2.4G2). Incubate on ice for 15 min. Centrifuge at 1500 RPM for 10 minutes at 8˚C and pellet.
6. Remember to save some cells to stain single color controls for the set-up of the machine. You must have enough unstained cells and cells stained with each color used in order to set up the machine. Preferably use brightly staining reagents that stain a high percentage of cells, such as B220 or Thy1 for mice.
7. Add antibodies to stain the cells that will be sorted; use at 1x or 0.5x of the typical concentration used to stain cells for analysis. Incubate for 20-30 min on ice. Fill tube with media up to top. Spin 10 min. @ 1500 RPM, 8˚C, remove supernatant and re-suspend pellet.
8. Stain with secondary reagent, if needed, for 20 min. on ice. Wash as before.
9. Wash once more with Sorting Buffer. Cells must be in low protein buffer (low FCS or BSA) to prevent the sorters from clogging.
10. Re-suspend cells at a concentration of 20-50×10^6/ml. This will ensure a sorting speed of approximately 18,000-20,000 events per second at the optimal pressure. Filter again through 70uM filter, again to prevent clogging.
11. Collection tubes can be blocked with FCS prior to adding collection medium. Collection medium can be supplemented with additional serum to offset the sheath buffer that will dilute the collection medium as cells are collected. Optimal conditions need to be worked out individually depending on the cells sorted, etc. Cells can be collected in either FACS tubes or 15 mL conical tubes.
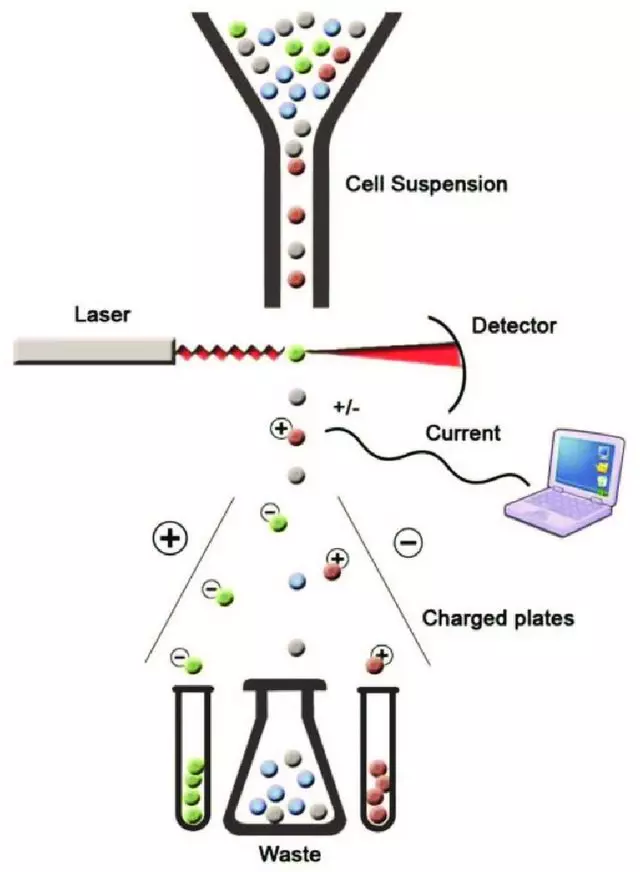
Principle of fluorescence-activated cell sorting (FACS) (Adopted from: Mehanna, Radwa. (2017). Physical versus Immunological Purification of Mesenchymal Stem Cells. 10.5772/intechopen.69295.)






 Guidence of GeneMedi's protocol / procedure for the diagnostics application:
Guidence of GeneMedi's protocol / procedure for the diagnostics application: 