SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit (Column-based) (Cat No.:GM-CD14-Cell-iso-kit)
Isolate pristine Human Classical Monocytes (CD14++) with the SOLIDEX™-ISOEx Kit. Utilizing a superior Column-based Negative (Untouched) selection strategy, this kit depletes non-target cells from PBMC using optimized Magnetic Beads. The eluted cells are free of antibodies and beads, ensuring Zero Activation and preserving their native phenotype. Achieve High Purity and high viability for sensitive downstream applications like macrophage differentiation.
SOLIDEX™-ISOEx Untouched Human Classical Monocyte Cell Isolation Kit (Column-based) is a Indirect CD14+ Cell Isolation Kit (Column-based, Non-releasable)
Check GeneMedi's SOLIDEX™-ISOEx
Cell Isolation Kit & Nanobeads
Order information
| Catalog No. | Product Name | Size | Price(In USD) |
|---|---|---|---|
| GM-CD14-Cell-iso-kit-10T | SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit (Column-based) | 10 Tests | 190 |
| GM-CD14-Cell-iso-kit-50T | SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit (Column-based) | 50 Tests | 570 |
| GM-CD14-Cell-iso-kit-100T | SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit (Column-based) | 100 Tests | 1004 |
| GM-CD14-Cell-iso-kit-750T | SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit (Column-based) | 750 Tests |
Description
| Cat No. | GM-CD14-Cell-iso-kit |
| Product Name | SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit (Column-based) |
| Description | SOLIDEX™-ISOEx Untouched Human Classical Monocyte Cell Isolation Kit (Column-based) is a Indirect CD14+ Cell Isolation Kit (Column-based, Non-releasable) |
| Application | SOLIDEX™-ISOEx Untouched Human Classical Monocyte Cell Isolation Kit (Column-based) is used for Negative CD14+ Cell Isolation (Untouched), with the Key Features, such as Preserves native cell function; maintains surface marker integrity; ideal for functional studies |
| Component for 5E8 cells (50Tests) / 1E9 cells(100tests) / 7.5E9 cells (750tests) |
GM-Tg-hg-T23212-iso-Cocktail-1-50μL/250μL/500μL/3.75mL: -50μL/250μL/500μL/3.75mL GMP-SMT-219-Ab01-nanoIMB-300μL/1.5ml/3ml/22.5ml: SOLIDEX™-ISOEx anti-Biotin Nanobeads (Column-Based)-300μL/1.5ml/3ml/22.5ml |
| Labeling Type | Indirect |
| Bead Type | Non-releasable |
| Format | Column-based |
| Key feature | Preserves native cell function; maintains surface marker integrity; ideal for functional studies |
| Cell population | CD14+CD16- Cell |
| Storage | Store at 2℃-8℃ |
Application
- Differentiation Studies: Generation of monocyte-derived macrophages (M1/M2) or dendritic cells (Mo-DCs) in vitro.
- Innate Immunity Research: Analysis of TLR signaling pathways and cytokine secretion profiling.
- Single-Cell Sequencing (scRNA-seq): High-quality, stress-free cells minimize transcriptomic artifacts caused by activation.
- Migration and Chemotaxis Assays: utilizing native, unblocked receptors.
Protocol
- Preparation: Prepare a single-cell suspension from cryopreserved or fresh PBMCs.
- Labeling: Add the Biotin-Antibody Cocktail (targets non-CD14 cells: T cells, B cells, NK cells, etc.). Incubate.
- Bead Binding: Add Anti-Biotin Microbeads to bind the labeled non-target cells. Incubate.
- Separation: Apply the suspension to the separation column placed in a magnetic field.
- Elution: Unlabeled CD14+ Classical Monocytes pass through the column freely and are collected (Untouched).
Validation Data
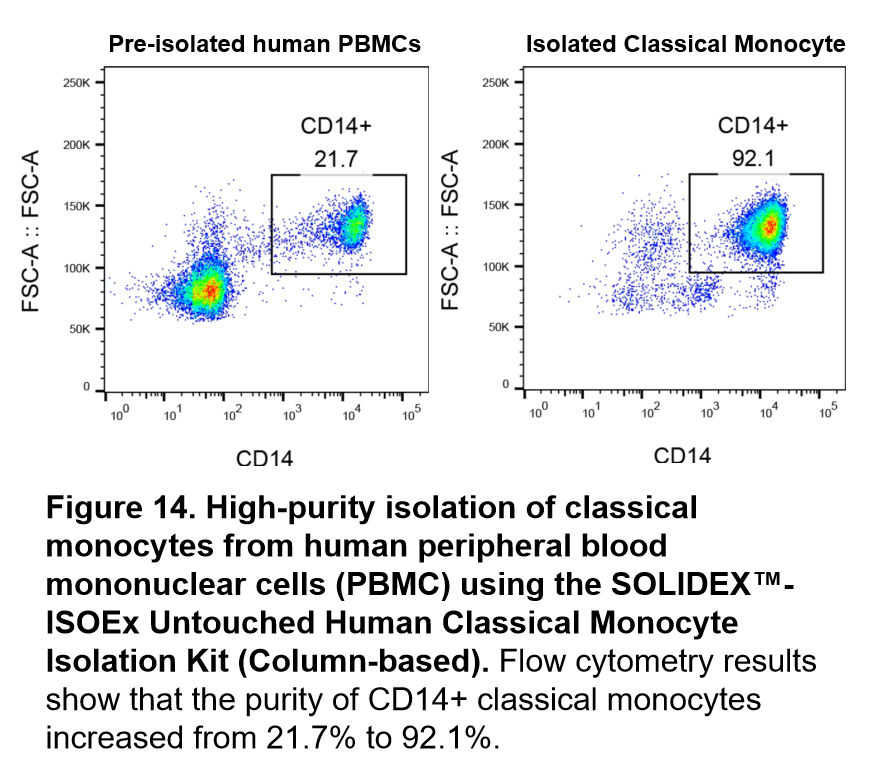
High-purity isolation of classical monocytes from human peripheral blood mononuclear cells (PBMC) using the SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit (Column-based)
Catalog No. GM-CD14-Cell-iso-kit
In this experiment, the SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit (Column-based) was used to isolate and purify classical monocytes from cryopreserved PBMCs. First, non-monocytic cells were specifically labeled with a biotin-conjugated antibody cocktail (Antibody Cocktail), followed by incubation with anti-biotin nano-magnetic beads. The purity of the cell populations before and after isolation was analyzed by flow cytometry. The purity of CD14+ classical monocytes in the flow-through fraction increased from 21.7% to 92.1%. The experimental results demonstrate that this kit effectively removes contaminating cells, yielding high-purity, untouched (antibody-unbound) classical monocytes.
Figure 14. High-purity isolation of classical monocytes from human peripheral blood mononuclear cells (PBMC) using the SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit (Column-based). Flow cytometry results show that the purity of CD14+ classical monocytes increased from 21.7% to 92.1%.
Click to get more Data / Case study about the product.
Background
Why Choose SOLIDEX™-ISOEx Untouched Human Classical Monocyte Isolation Kit?
- True "Untouched" Isolation: Target monocytes are never bound by antibodies or beads, preventing Fc-receptor activation and preserving differentiation potential.
- Exceptional Purity: The Column-based matrix acts as a physical filter, allowing rigorous washing of contaminants to achieve >90% purity compared to column-free methods.
- Preserved Physiology: Gentle isolation ensures cells remain in a resting state, critical for accurate immunological studies.
- Rapid Workflow: Isolate cells in under 45 minutes with a simple, standardized protocol.
FAQ
What are the sample requirements?
Can I stain the cells for Flow Cytometry immediately after isolation?
How does the Column-based separation work?
Are the separation columns reusable?
What is the advantage of Column-based over Column-free methods?
Is this protocol compatible with automated systems?






