SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based) (Cat No.:GM-CD4-T-Cell-iso-kit)
Maximize your research potential with GeneMedi’s SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit. Specifically designed for Human CD4+ T Cell isolation from PBMC, this Column-based system employs a precise Untouched selection strategy.
SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based) is a Indirect CD4+ T Cell Isolation Kit (Column-based, Non-releasable)
Check GeneMedi's SOLIDEX®-ISOEx
Cell Isolation Kit & Nanobeads
Order information
| Catalog No. | Product Name | Size | Price(In USD) |
|---|---|---|---|
| GM-CD4-T-Cell-iso-kit-10T | SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based) | 10 Tests | 190 |
| GM-CD4-T-Cell-iso-kit-50T | SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based) | 50 Tests | 570 |
| GM-CD4-T-Cell-iso-kit-100T | SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based) | 100 Tests | 1004 |
| GM-CD4-T-Cell-iso-kit-750T | SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based) | 750 Tests |
Description
| Cat No. | GM-CD4-T-Cell-iso-kit |
| Product Name | SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based) |
| Description | SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based) is a Indirect CD4+ T Cell Isolation Kit (Column-based, Non-releasable) |
| Application | SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based) is used for Negative CD4+ T Cell Isolation (Untouched), with the Key Features, such as Preserves native cell function; maintains surface marker integrity; ideal for functional studies |
| Component for 5E8 cells (50Tests) / 1E9 cells(100tests) / 7.5E9 cells (750tests) |
GM-CD4-T-Cell-iso-Cocktail-1-50μL/250μL/500μL/3.75mL: SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Cocktail-50μL/250μL/500μL/3.75mL GMP-SMT-219-Ab01-nanoIMB-300μL/1.5ml/3ml/22.5ml: SOLIDEX®-ISOEx anti-Biotin Nanobeads (Column-Based)-300μL/1.5ml/3ml/22.5ml |
| Labeling Type | Indirect |
| Bead Type | Non-releasable |
| Format | Column-based |
| Key feature | Preserves native cell function; maintains surface marker integrity; ideal for functional studies |
| Cell population | CD4-T Cell |
| Storage | Store at 2°C–8°C |
Application
- Helper T Cell differentiation assays: Ideal for studying Th1, Th2, Th17, and Treg polarization.
- HIV/Viral Research: High-purity CD4+ cells for studying viral entry and replication mechanisms.
- Co-culture studies: Assessment of T cell-B cell interactions or macrophage activation.
- Epigenetic and Transcriptomic analysis: Pristine RNA/DNA extraction for ATAC-seq or RNA-seq.
Protocol
- Sample Prep: Isolate PBMCs and resuspend in separation buffer.
- Labeling: Add the isolation cocktail to the sample and incubate for 5 minutes at room temperature.
- Bead Incubation: Add Anti-biotin Nanobeads and incubate for 10 minutes at 2-8°C.
- Separation: Apply the cell suspension to an LS Column placed in a magnetic field. Collect the flow-through, which contains the enriched, untouched CD4+ T cells.
Validation Data
Efficient Enrichment of High-Purity CD4+ T Cells from Human PBMCs Using SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based)
Catalog No. GM-CD4-T-Cell-iso-kit
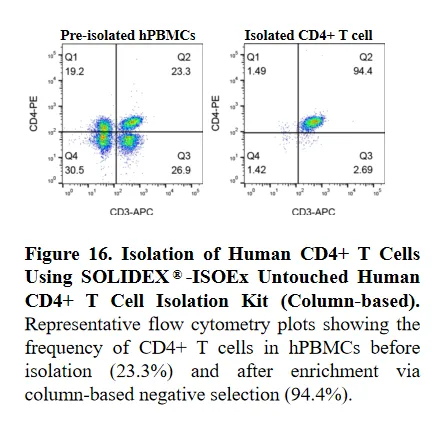
To evaluate the isolation efficiency of the SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based), CD4+ T cells were enriched from human peripheral blood mononuclear cells (hPBMCs) via negative selection. Non-target cells were labeled with a biotinylated antibody cocktail and depleted using magnetic beads within a separation column.
Cell fractions were stained with CD3-APC and CD4-PE for flow cytometric analysis. The frequency of CD4+ T cells demonstrated a substantial increase from 23.3% in the pre-isolation sample to 94.4% in the post-isolation fraction. These results confirm that the column-based system provides superior depletion of non-target populations, yielding highly purified "untouched" CD4+ T cells.
Figure 16. Isolation of Human CD4+ T Cells Using SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit (Column-based).Representative flow cytometry plots showing the frequency of CD4+ T cells in hPBMCs before isolation (23.3%) and after enrichment via column-based negative selection (94.4%).
Click to get more Data / Case study about the product.
Background
Why Choose SOLIDEX®-ISOEx Untouched Human CD4+ T Cell Isolation Kit?
- Zero Activation: The "Untouched" negative selection ensures CD4 receptors are not engaged by antibodies, preventing artificial signaling.
- Superior Purity: The column-based matrix enhances separation efficiency, typically achieving higher purity compared to column-free methods.
- Rapid Workflow: Complete the labeling and separation process in approximately 20 minutes.
- Downstream Ready: Cells are immediately ready for flow cytometry staining or culture without bead interference.
FAQ
What are the sample requirements?
Can I stain the cells for Flow Cytometry immediately after isolation?
How does the column-based negative selection work?
Can the columns be reused?
What is the advantage of this column-based method over column-free methods?
Is this workflow compatible with automation?