SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Kit (Column-based) (Cat No.:GM-Pan-T-Cell-iso-kit)
Achieve superior purity with GeneMedi’s SOLIDEX®-ISOEx Untouched Human Pan-T Cell Isolation Kit. Optimized for Human Pan-T Cell isolation from PBMC, this Column-based system utilizes a negative selection strategy to deliver truly "untouched" cells. Our biotin-conjugated antibody cocktail and Magnetic Beads remove non-T cells, ensuring the target CD3+ cells remain free of antibodies and bead residues. With High Purity (>95%) and zero activation, these cells are ideal for downstream functional assays.
SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Kit (Column-based) is a Indirect CD3+ Pan-T Cell Isolation Kit (Column-based, Non-releasable)
Check GeneMedi's SOLIDEX®-ISOEx
Cell Isolation Kit & Nanobeads
Order information
| Catalog No. | Product Name | Size | Price(In USD) |
|---|---|---|---|
| GM-Pan-T-Cell-iso-kit-10T | SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Kit (Column-based) | 10 Tests | 190 |
| GM-Pan-T-Cell-iso-kit-50T | SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Kit (Column-based) | 50 Tests | 570 |
| GM-Pan-T-Cell-iso-kit-100T | SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Kit (Column-based) | 100 Tests | 1004 |
| GM-Pan-T-Cell-iso-kit-750T | SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Kit (Column-based) | 750 Tests |
Description
| Cat No. | GM-Pan-T-Cell-iso-kit |
| Product Name | SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Kit (Column-based) |
| Description | SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Kit (Column-based) is a Indirect CD3+ Pan-T Cell Isolation Kit (Column-based, Non-releasable) |
| Application | SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Kit (Column-based) is used for Negative CD3+ Pan-T Cell Isolation (Untouched), with the Key Features, such as Preserves native cell function; maintains surface marker integrity; ideal for functional studies |
| Component for 5E8 cells (50Tests) / 1E9 cells(100tests) / 7.5E9 cells (750tests) |
GM-Pan-T-Cell-iso-Cocktail-1-50μL/250μL/500μL/3.75mL: SOLIDEX®-ISOEx Untouched Human CD3+ Pan-T Cell Isolation Cocktail-50μL/250μL/500μL/3.75mL GMP-SMT-219-Ab01-nanoIMB-200μL/1ml/2ml/15ml: SOLIDEX®-ISOEx anti-Biotin Nanobeads (Column-Based)-200μL/1ml/2ml/15ml |
| Labeling Type | Indirect |
| Bead Type | Non-releasable |
| Format | Column-based |
| Key feature | Preserves native cell function; maintains surface marker integrity; ideal for functional studies |
| Cell population | CD3+ Pan-T Cell |
| Storage | Store at 2°C–8°C |
Application
- In vitro T cell activation and expansion: Perfect for functional studies as cells are unstimulated.
- Flow Cytometry analysis: Cells have no surface antibodies, allowing immediate staining with structural integrity.
- Molecular biology assays: High purity RNA/DNA extraction for sequencing or PCR.
- Adoptive cell therapy research: Starting material for CAR-T or TCR-T development.
Protocol
- Preparation: Isolate and count PBMCs; resuspend in separation buffer.
- Labeling: Add the Isolation Cocktail to cells and incubate for 5 minutes at room temperature.
- Magnetic Binding: Add Anti-biotin Nanobeads and incubate for 10 minutes at 2-8°C.
- Separation: Apply cell suspension to an LS Column on a magnetic separator. Collect the flow-through (this contains your enriched Pan-T cells).
Validation Data
High-purity isolation of Pan-T cells from human PBMC using the SOLIDEX®-ISOEx Untouched Human Pan-T Cell Isolation Kit (Column-based)
Catalog No. GM-Pan-T-Cell-iso-kit
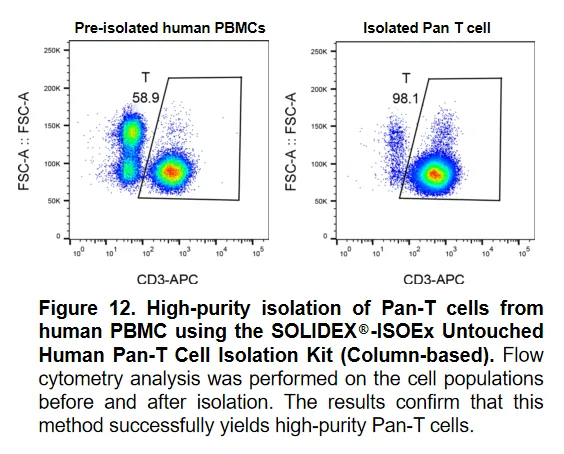
In this experiment, the SOLIDEX®-ISOEx Untouched Human Pan-T Cell Isolation Kit (Column-based) was used to isolate and purify Pan-T cells from human peripheral blood mononuclear cells (PBMC). First, non-Pan-T cells were specifically labeled with a biotin-conjugated antibody cocktail (Antibody Cocktail), followed by incubation with anti-biotin nano-magnetic beads. When the cell suspension passed through the separation column, the magnetically labeled non-target cells were retained in the column, while the unlabeled, untouched Pan-T cells flowed out with the elution buffer. Data showed that the proportion of Pan-T cells increased significantly from 58.9% before isolation to 96.1% after isolation. The experimental results demonstrate that this kit effectively removes various types of non-Pan-T cells, yielding high-purity Pan-T cells that retain their original biological activity.
Figure 12. High-purity isolation of Pan-T cells from human PBMC using the SOLIDEX®-ISOEx Untouched Human Pan-T Cell Isolation Kit (Column-based). Flow cytometry analysis was performed on the cell populations before and after isolation. The results confirm that this method successfully yields high-purity Pan-T cells.
Click to get more Data / Case study about the product.
Background
Why Choose SOLIDEX®-ISOEx Untouched Human Pan-T Cell Isolation Kit?
- Truly Untouched: Uses negative selection to leave target T cells completely free of antibodies and magnetic particles, preserving their native function.
- High Purity: Validated to achieve >98% purity (compared to ~58% in PBMCs).
- Fast Workflow: Optimized rapid protocol with only 15 minutes of total incubation time.
- High Compatibility: Compatible with standard LS columns and magnetic separators.
FAQ
What are the sample requirements?
Can I stain the cells for Flow Cytometry immediately after isolation?
How does the column-based negative selection work?
Can the columns be reused?
What is the advantage of this column-based method over column-free methods?
Is this workflow compatible with automation?