COVID-19 Guidance and information collection on the new mutant variants
COVID-19 Guidance and information collection on the new mutant variants of the SARS-CoV-2 (2019nCoV) virus
- COVID-19 (SARS-CoV-2): information about the new virus variant
- New SARS-COV-2 variant: information and risk assessment
- Central Alerting System (CAS) alert
- New variant clustering in households analysis (ONS)
- SARS-CoV-2 lateral flow antigen tests: evaluation of VUI-202012/01
- WTO: Statement of the WHO Working Group on COVID-19 Animal Models (WHO-COM) about the UK and South African SARS-CoV-2 new variants.
- EMA guidance for COVID-19 vaccine
- Investigation of novel SARS-CoV-2 variant: Variant of Concern 202012/01
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 6
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 5
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 4
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 3
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 2
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 1
New variant of SARS-COV-2 (2019nCOV) B.1.1.7 lineage spreaded in UK
The world is in midst of the COVID-19 pandemic. Recently a novel SARS-COV-2 (2019nCOV) lineage, the B.1.1.7 lineage, with serials of site mutation, shows stronger infection ability in the UK. The SARS-COV-2 B.1.1.7 lineage carries a larger than a usual number of coronavirus genetic changes.Extended Reading: Preliminary genomic characterisation of an emergent SARS-CoV-2 lineage in the UK defined by a novel set of spike mutations
|
Relative products collection: GeneMedi products for New variant of SARS-COV-2 (2019nCOV) UK B.1.1.7 lineage |
New variant of SARS-COV-2 (2019nCOV) 501Y.V2 lineage(B.1.351) spreaded in South African SARS-CoV-2
The South African variant is characterized by eight lineage-defining mutations in the spike protein including three key residues in the receptor binding domain (K417N, E484K and N501Y) and is referred to as lineage 501Y.V2.Extended Reading: Alert Notification: New SARS-CoV-2 variant with multiple spike protein mutations
|
Relative products collection: GeneMedi products for New variant of SARS-COV-2 (2019nCOV) South Africa 501Y.V2 lineage(B.1.351) |
About COVID-19 Pandemic and SARS-CoV-2 Vaccine
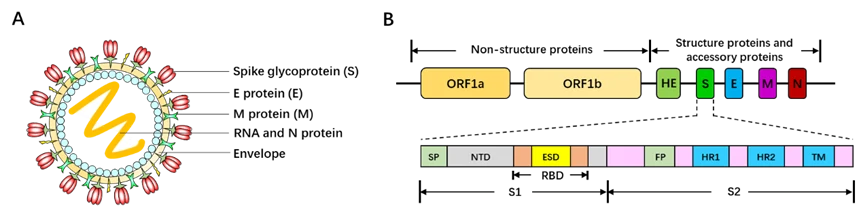
Coronavirus Disease 2019 (COVID-19) is a novel viral pneumonia caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). First discovered in Wuhan, a city in Hubei province of China, COVID-19 has already broken out throughout the world and posed a great threat to the public health, especially in Europe and North America now. Additionally, person-to-person transmission of COVID-19 disease is reported to be extremely rapid [158-160]. To date, more than one million cases were infected with COVID-19 and over 55,000 deaths occurred. Therefore, it is really urgent and noteworthy to develop the vaccines specific to COVID-19/SARS-CoV-2.Belonging to the Betacoronavirus genus family, SARS-CoV-2 is 60~200nm in diameter and encapsidates a large positive-sense, single-stranded RNA virus (26-32kb) with many spikes on the virus capsid (Fig. 17A). The RNA genome of SARS-CoV-2 encodes several accessory proteins and structural proteins, such as nucleocapsid (N) protein, envelope (E) protein, membrane (M) protein, and spike (S) protein (Fig. 10B). Although the detailed mechanism of SARS-CoV-2 infection has not been clearly illuminated, several studies demonstrated that SARS-CoV-2 enters human cells via utilizing spike (S) protein to bind to the angiotensin converting enzyme (ACE2) on the surface of target cell [161, 162].
1. 2 Korber, B. et al. Spike mutation pipeline reveals the emergence of a more transmissible form of SARS-CoV-2. bioRxiv Preprint., doi:10.1101/2020.04.29.069054 (2020). 2. Investigation of novel SARS-COV-2 variant. Public health England. 3. European Centre for Disease Prevention and Control. Rapid increase of a SARS-CoV-2 variant with multiple spike protein mutations observed in the United Kingdom – 20 December 2020.ECDC: Stockholm; 2020. 4. Li, G. & De Clercq, E. Therapeutic options for the 2019 novel coronavirus (2019-nCoV). Nature Reviews Drug Discovery, doi:10.1038/d41573-020-00016-0 (2020). 5. Haque, A. & Pant, A. B. Efforts at COVID-19 Vaccine Development: Challenges and Successes. Vaccines 8, doi:10.3390/vaccines8040739 (2020). 6. Dong, Y. et al. A systematic review of SARS-CoV-2 vaccine candidates. Signal Transduction and Targeted Therapy 5, doi:10.1038/s41392-020-00352-y (2020). 7. Andrew Rambaut1, N. L., Oliver Pybus, Wendy Barclay, Jeff Barrett5, Alesandro Carabelli6, Tom Connor, Tom Peacock, David L Robertson8, Erik Volz, . Preliminary genomic characterisation of an emergent SARS-CoV-2 lineage in the UK defined by a novel set of spike mutations. https://virological.org/ (2020). 8. Rapid increase of a SARS-CoV-2 variant with multiple spike protein mutations observed in the United Kingdom. European Centre for Disease Prevention and Control: Publications & data. 9. Zhang, L. et al. SARS-CoV-2 RNA reverse-transcribed and integrated into the human genome. bioRxiv Preprint. , doi:10.1101/2020.12.12.422516 (2020).






 PSV validated Data Poster Download
PSV validated Data Poster Download