Products & solutions & information collection of COVID-19 vaccines, new variants of SARS-CoV-2 and efficacy evaluation solutions.
|
Content index Background: Products & Procotol Collection: 6. Validated SARS-CoV-2 neutralizing antibodies Information Collection: 1. Landscape of global COVID-19 vaccine candidates development 2. COVID-19 Guidance and information collection on the new mutant variants 3. COVID-19 News and announcements collection on the new mutant variants 4. About COVID-19 Pandemic and SARS-CoV-2 Vaccine Reference Click to check>> |
BACKGROUND
Challenge of COVID-19 vaccine discovery & development: to meet accumulated mutating of SARS-CoV-2 and a long-term viral genome transcrption
COVID-19 (Coronavirus Disease 2019) is novel viral pneumonia caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2, also called 2019nCoV). The world is in midst of the COVID-19 pandemic. Effective vaccines are needed to halt the spread of the SARS-CoV-2 pandemic. Based on the data from WTO, there are 52 COVID-19 vaccines in clinical stage (Supplementary Table 1.) and 162 COVID-19 vaccine candidates in pre-clinical stage (Supplementary Table 2.) 2 mRNA vaccines, the BNT162b2 from Pfizer&BioNTech and the mRNA-1273 from Moderna, have recently been issued the emergency use authorization (EUA) by the U.S. Food and Drug Administration (FDA) for the prevention of COVID-19 in individuals 16 years of age and older.
The efficacy and safety of COVID-19 vaccine candidates need carefully investigated, from pre-clinical, clinal stage to a long time after BLA. There are 2 important items here:
Firstly, the coronavirus is kept accumulated mutating. The most important mutation occurs in SARS-CoV-2 (2019nCoV) Spike protein (SARS-CoV-2 S protein). The SARS-CoV-2 (2019nCoV) Spike mediates binding and entry into host cells and is a major target of neutralizing antibodies. Most of the COVID-19 vaccines focus on spike protein1,4-6.
Different SARS-CoV-2 lineages with diverse Spike protein mutant variants may yield a heavy impact on the course of the pandemic2-3. The United Kingdom (UK) has faced a rapid increase in COVID-19 cases caused by a novel SARS-COV-2 (2019nCOV) lineage, the B.1.1.7 lineage, which carries a larger than a usual number of coronavirus genetic changes7-8, particularly in the SARS-CoV-2 spike protein(Table 1)7.
Table 1 | All mutations and deletions occur in COVID-19 SARS-COV-2 (2019nCOV) B.1.1.7 lineage
(Click to more details about B.1.1.7 lineage related products).
| Gene | Nucleotide | Amino acid | |
| ORF1ab | C3267T | T1001I | C5388A | A1708D | T6954C | I2230T | 11288-11296 deletion | SGF 3675-3677 deletion |
| spike | 21765-21770 deletion | HV 69-70 deletion (Click to more details about HV 69-70 deletion related products) | 21991-21993 deletion | Y144 deletion | A23063T | N501Y (Click to more details about N501Y related products) | C23271A | A570D | C23604A | P681H | C23709T | T716I | T24506G | S982A | G24914C | D1118H |
| Orf8 | C27972T | Q27stop | G28048T | R52I | A28111G | Y73C |
| N | 28280 GAT->CTA | D3L | C28977T | S235F |
Secondly, SARS-CoV-2 was reported to integrate into the genome, which means a stable long-term expression of transcript products of SARS-CoV-2 9.
In conclusion, the Efficacy of COVID-19 vaccines and neutralized antibodies need long-term tracking, which mainly focuses on the novel spike protein mutation occurring in the new lineage variant of SARS-CoV2. The efficacy evaluation solution and tools of COVID-19 vaccines and neutralized antibodies need continually updated.
PRODUCTS & PROCOTOL COLLECTION
GeneMedi Efficacy Assay/Evaluation Solutions for COVID-19 Vaccines and Therapeutic antibodies against SARS-CoV2(2019nCoV)
To meet accumulated mutating of SARS-CoV-2, GeneMedi keeps continually developing novel solutions and tools for efficacy evaluation of COVID-19 vaccines (and neutralized antibodies) against novel mutant SARS-CoV-2 (2019nCoV) lineages. Our scientist's team takes duty in fighting against the COVID-19 pandemic with our global industrial and academic partners.
1. GeneMedi information and products collection of SARS-COV-2 (2019nCOV) UK B.1.1.7 lineage and South Africa 501Y.V2 lineage and Brazilian P.1 lineage(B.1.1.28.1)
GeneMedi pseudotyped virus (pseudovirus) of SARS-COV-2 (2019nCOV) B.1.1.7 lineage
GeneMedi pseudotyped virus (pseudovirus) of SARS-COV-2 (2019nCOV) 501Y.V2 lineage
GeneMedi pseudotype virus (pseudovirus) of SARS-COV-2 (2019nCOV) P.1 lineage(B.1.1.28.1, Brazilian variant)
SARS-COV-2 (2019nCOV) B.1.1.7 lineage & 501Y.V2 lineage(B.1.351) & P.1 lineage (B.1.1.28.1, Brazilian variant) of Spike protein & ACE2 competition binding assay for efficacy evaluation of COVID-19 vaccines and therapeutic antibodies
GeneMedi codon-optimized spike mammalian expression vector for SARS-COV-2 (2019nCOV) B.1.1.7 lineage
GeneMedi codon-optimized spike mammalian expression vector for SARS-COV-2 (2019nCOV) South Africa 501Y.V2 lineage(B.1.351).
GeneMedi codon-optimized spike mammalian expression vector for SARS-COV-2 (2019nCOV) - P.1 lineage(B.1.1.28.1, Brazilian variant)
2. Multiple variants of Spike protein & ACE2 competition binding assay for efficacy evaluation of COVID-19 vaccines and therapeutic antibodies
The Spike proteins variants that GeneMedi offer is including:
1) Multiple variants of recombinant Spike protein for Spike protein & ACE2 competition binding assay for efficacy evaluation of COVID-19 vaccines and therapeutic antibodies
| Cat No. | Antigen Name of 2019-nCoV(SARS-CoV-2) | Isotypes | Order |
| GMP-H-ACE2001 | Recombinant human solubale ACE2 protein (soluble hACE2, extracellular hACE2, C-His) | Mammalian (human cell) |  |
| GMP-H-ACE2002 | Recombinant human solubale ACE2 protein (soluble hACE2, extracellular hACE2, C-His) | Mammalian (human cell) |  |
- Competition assay for neutralizing antibodies, peptides inhibitor and vaccines-immunized serums
3. Multi-VariantsTM SARS-CoV2 (wildtype, D614G, N501Y, E484K, B.1.1.7 lineage, 501Y.V2 lineage, and so on) Pseudotype virus-Based Neutralization Assay System for efficacy evaluation of COVID-19 vaccines and therapeutic antibodies (Lentiviral pseudovirus)
|
Protocol: GeneMedi SARS-CoV2 pseudovirus-based neutralization assay-protocol |
4. Codon-optimized mammalian expression vector for SARS-COV-2 (2019nCOV) spike wide type & mutant variants (D614G, N501Y, E484K, B.1.1.7 lineage, 501Y.V2 lineage, and so on)
Codon-optimized viral particle (Adenovirus, Lentivirus, AAV) for SARS-COV-2 (2019nCOV) spike wide type & mutant variants (D614G, N501Y, E484K, B.1.1.7 lineage, 501Y.V2 lineage, and so on)
5. 293T-ACE2 stable cell line & human ACE2 expression vectors: Effector cell of pseudotype virus-Based Neutralization Assay System for efficacy evaluation of COVID-19 vaccines and therapeutic antibodies (Lentiviral pseudovirus)
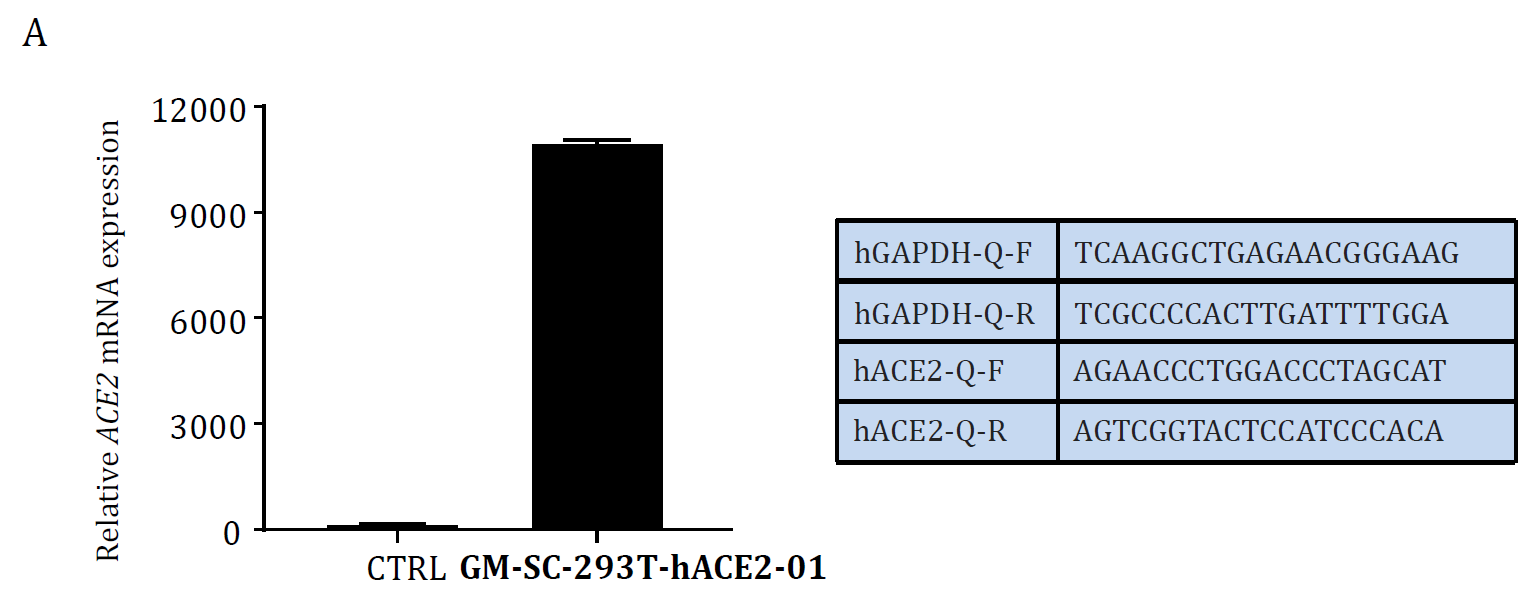
Human ACE2 overexpression stable HEK293T cell lines. Catalot Number: GM-SC-293T-hACE2-01 Click here for details
| Cat No. | 2019 nCoV related Gene |
Gene &Vector description of 2019 nCoV | Vector | Reporter | Tag | codon Optimized |
Order |
| GMV-V-2019nCoV-045 | TMPRSS2 | pGMLv-hTMPRSS2(C-3FLAG) | Lentiviral vector | Zsgreen | C-3FLAG | No |  |
| GMV-V-2019nCoV-041 | ACE2 | pGMLV-hACE2(C-3FLAG) | Lentiviral vector | Zsgreen | C-3FLAG | No |  |
| GMV-V-2019nCoV-042 | ACE2 | pAD-hACE2(C-3FLAG) | Pre-made Adenovirus | EGFP | C-3FLAG | No |  |
| GMV-V-2019nCoV-046 | TMPRSS2 | pGMLv-mtmprss2(C-3FLAG) | Lentiviral vector | Zsgreen | C-3FLAG | No |  |
| GMV-V-2019nCoV-043 | ACE2 | pAD-mACE2(C-3FLAG) | Pre-made Adenovirus | EGFP | C-3FLAG | No |  |
| GMV-V-2019nCoV-044 | ACE2 | pGMLV-mACE2(C-3FLAG) | Lentiviral vector | Zsgreen | C-3FLAG | No |  |
 |
|
|
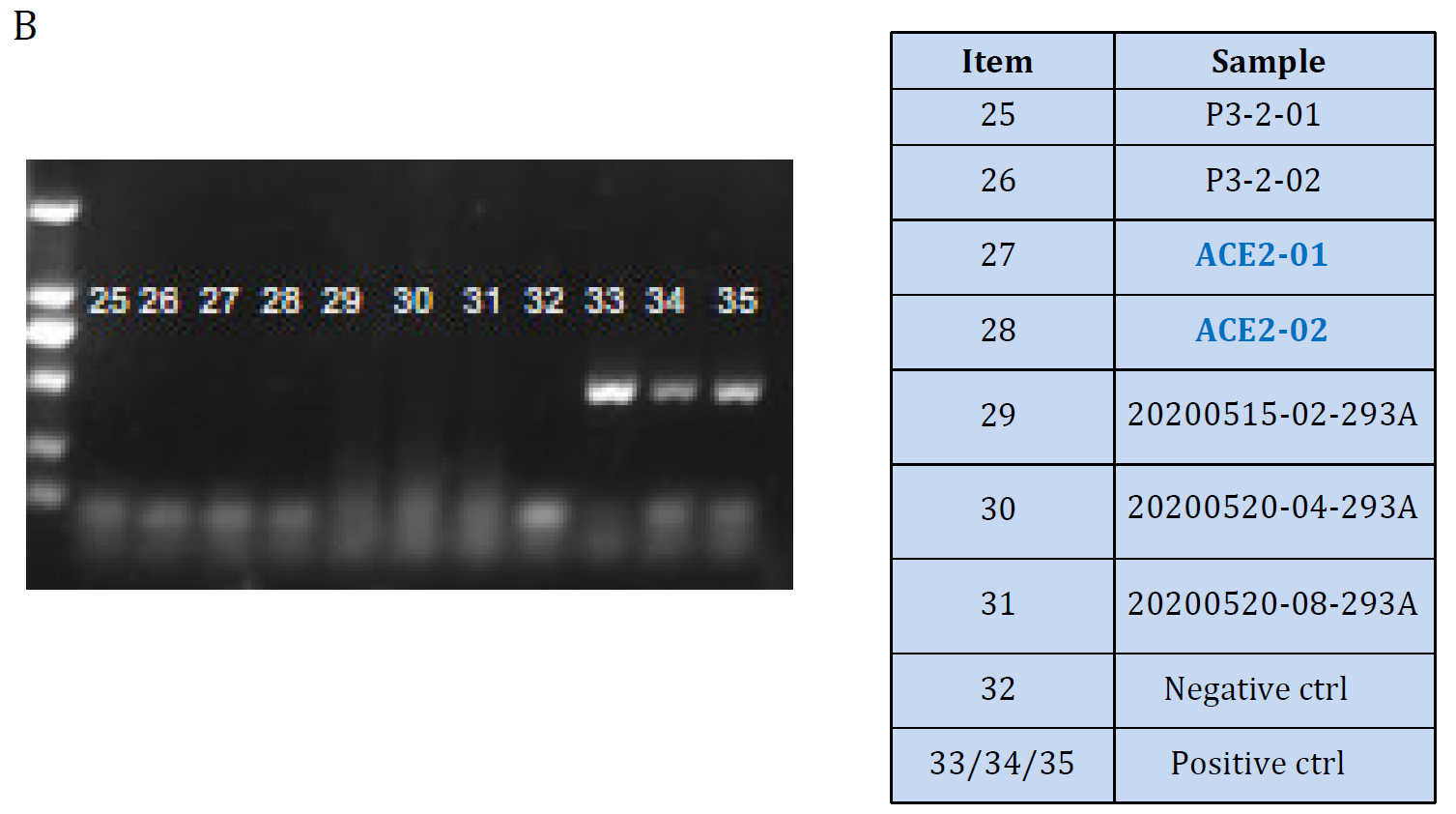
Figure. ACE2 mRNA level Validation in hACE2 overexpression stable HEK293T cell lines: Cat. GM‐SC‐293T‐hACE2‐01 (A) and the cell lines were tested as Mycoplasma free (B). |
|
| Cat No. | 2019 nCoV related Gene |
Gene &Vector description of 2019 nCoV | Vector | Reporter | Tag | codon Optimized |
Order |
| GMV-V-2019nCoV-041 | ACE2 | pGMLV-hACE2(C-3FLAG) | Lentiviral vector | Zsgreen | C-3FLAG | No |  |
| GMV-V-2019nCoV-042 | ACE2 | pAD-hACE2(C-3FLAG) | Pre-made Adenovirus | EGFP | C-3FLAG | No |  |
| GMV-V-2019nCoV-043 | ACE2 | pAD-mACE2(C-3FLAG) | Pre-made Adenovirus | EGFP | C-3FLAG | No |  |
| GMV-V-2019nCoV-044 | ACE2 | pGMLV-mACE2(C-3FLAG) | Lentiviral vector | Zsgreen | C-3FLAG | No |  |
6. Validated SARS-CoV-2 neutralizing antibodies-benchmark COVID-19 neutralizing antibodies.
| Cat No. | Antigen Name of 2019-nCoV(SARS-CoV-2) | Source (Expression Host) |
Isotypes | Bioactivity validation | Order |
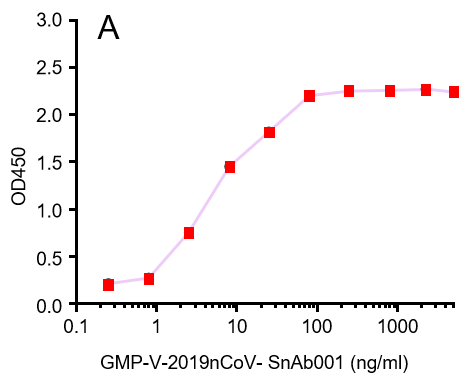
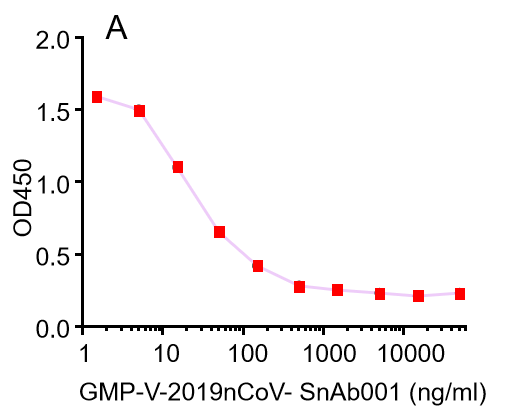
| GMP-V-2019nCoV-SnAb001 | Anti-2019-nCoV Spike (Spike RBD domain) human monoclonal neutralizing antibody (IgG1) | Mammalian (human cell) | human IgG1 | Validated in COVID-19 Spike protein and Spike-RBD protein binding affnity. COVID-19 related neutralizing potency is validated by 1.2019nCoV pseudotyped virus based neutralization assay in 293T-ACE2 effector cell. 2. competitively blocking the binding of ACE-2 receptor with SARS-CoV-2 Spike protein. |

|
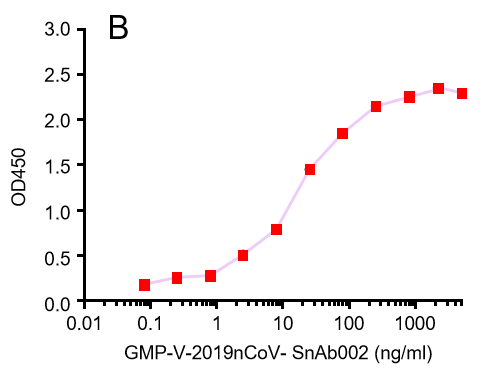
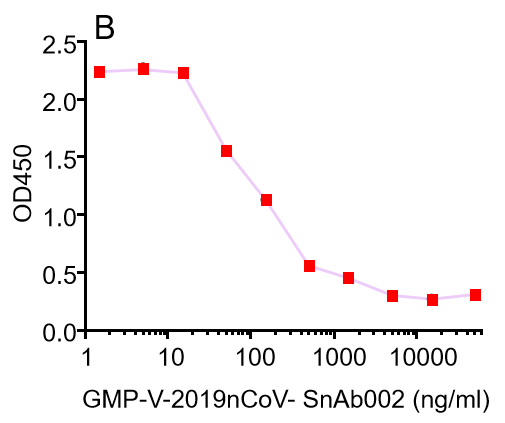
| GMP-V-2019nCoV-SnAb002 | Anti-2019-nCoV Spike (Spike RBD domain) human monoclonal neutralizing antibody (IgM) | Mammalian (human cell) | human IgM | Validated in COVID-19 Spike protein and Spike-RBD protein binding affnity. COVID-19 related neutralizing potency is validated by 1.2019nCoV pseudotyped virus based neutralization assay in 293T-ACE2 effector cell. 2. competitively blocking the binding of ACE-2 receptor with SARS-CoV-2 Spike protein. |

|
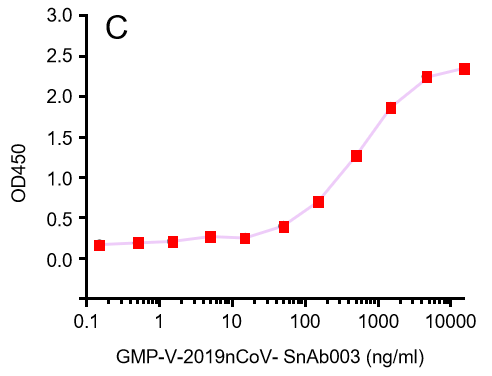
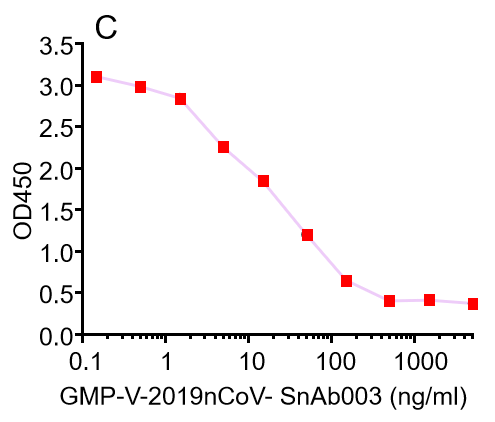
| GMP-V-2019nCoV-SnAb003 | Anti-2019-nCoV Spike (Spike RBD domain) human monoclonal neutralizing antibody (IgA) | Mammalian (human cell) | human IgA | Validated in COVID-19 Spike protein and Spike-RBD protein binding affnity. COVID-19 related neutralizing potency is validated by 1.2019nCoV pseudotyped virus based neutralization assay in 293T-ACE2 effector cell. 2. competitively blocking the binding of ACE-2 receptor with SARS-CoV-2 Spike protein. |

|
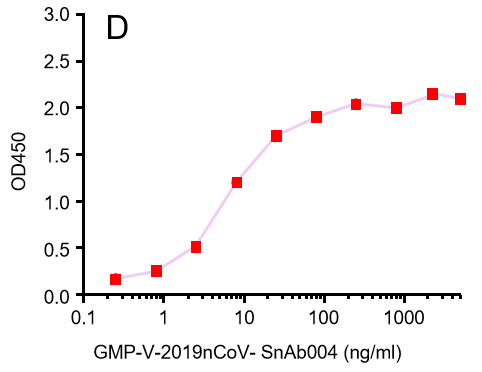
| GMP-V-2019nCoV-SnAb004 | Anti-2019-nCoV Spike (Spike RBD domain) mouse monoclonal neutralizing antibody (IgG1) | Mammalian (human cell) | mouse IgG1 | Validated in COVID-19 Spike protein and Spike-RBD protein binding affnity. COVID-19 related neutralizing potency is validated by 1.2019nCoV pseudotyped virus based neutralization assay in 293T-ACE2 effector cell. 2. competitively blocking the binding of ACE-2 receptor with SARS-CoV-2 Spike protein. |

|
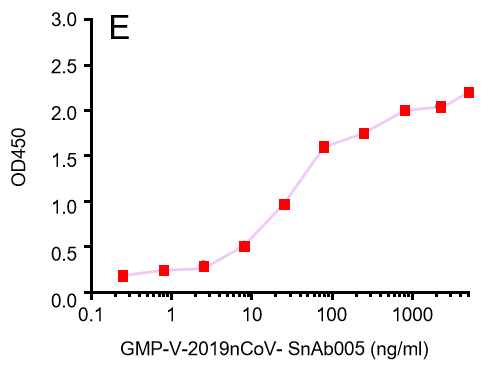
| GMP-V-2019nCoV-SnAb005 | Anti-2019-nCoV Spike (Spike RBD domain) Cynomolgus monoclonal neutralizing antibody (IgG1) | Mammalian (human cell) | Cynomolgus (Non human primate, NHP) IgG1 | Validated in COVID-19 Spike protein and Spike-RBD protein binding affnity. COVID-19 related neutralizing potency is validated by 1.2019nCoV pseudotyped virus based neutralization assay in 293T-ACE2 effector cell. 2. competitively blocking the binding of ACE-2 receptor with SARS-CoV-2 Spike protein. |

|
|
|
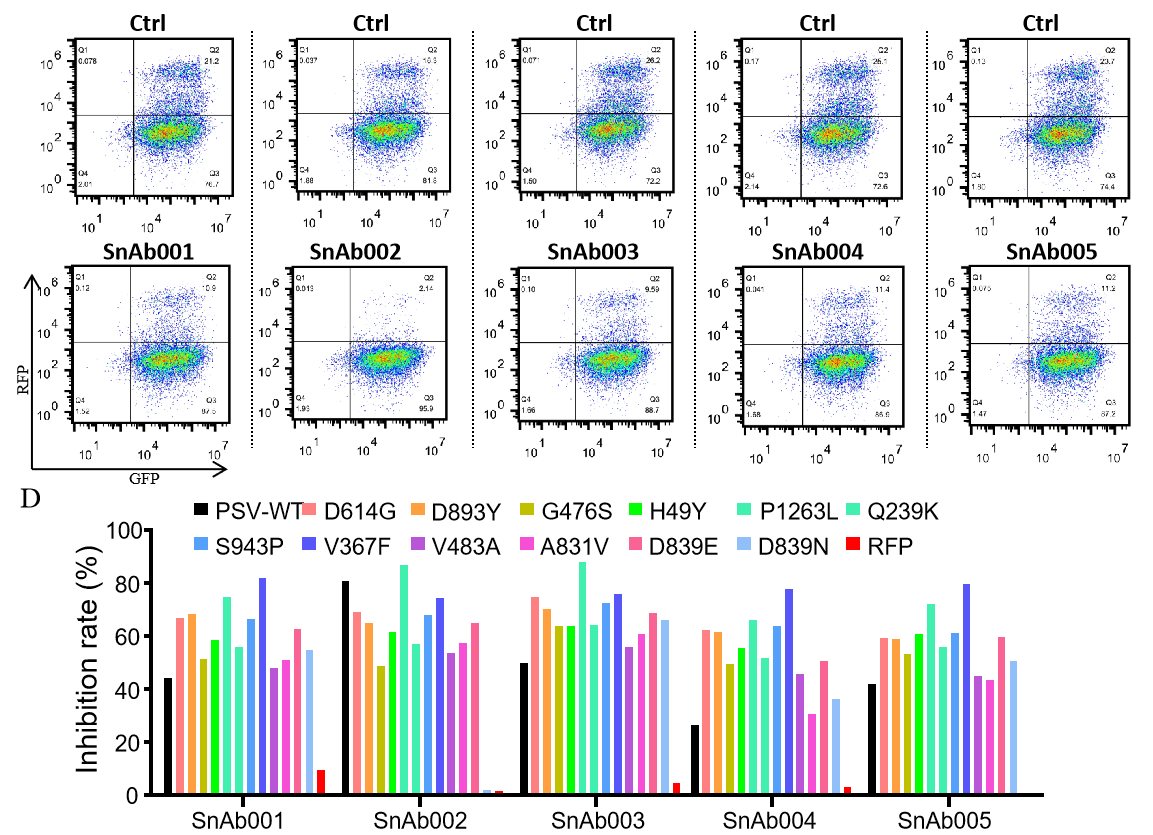
Figure. The Pseudovirus (PSV) Based Neutralizing Assay was performed on 293T-hACE2 cells infected with GeneMedi-SARS-CoV-2 WT and Spike Mutation Variants (D614G, S943P, V367F, G476S, V483A, H49Y, Q239K, A831V, P1263L, D839Y/N/E:D839Y, D839N, D839E) Pseudovirus (PSV) under treatment of GeneMedi's anti-2019-nCoV Spike Neutralizing antibodies (Nabs) . Inhibition rate was determined by comparing the relative RFP+GFP+/GFP+ rate. |
|
||||||||||||||||||
|
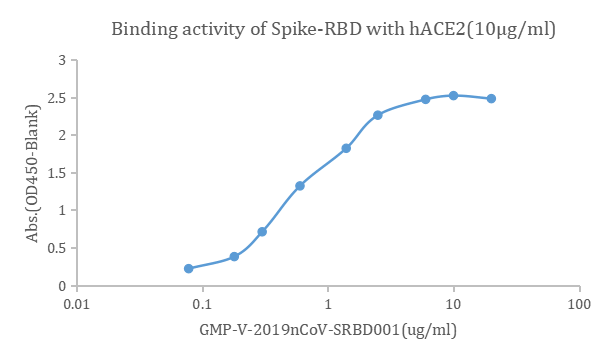
Figure. The binding of GeneMedi's anti-2019-nCoV Spike Neutralizing antibodies (Nabs) to Recombinant 2019-nCoV(SARS-CoV-2) Spike RBD protein (GMP-V-2019nCoV-SRBD001) at 5.0ug/ml (100uL/well) was measured by ELISA. |
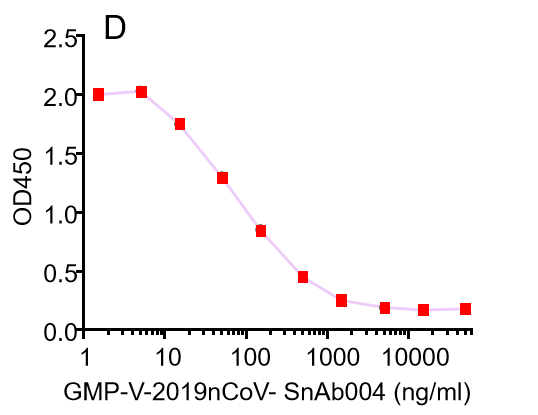
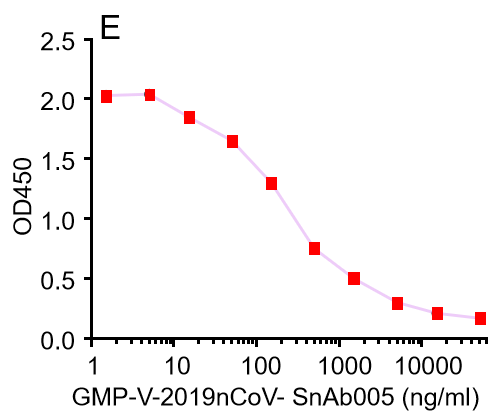
|
||||||||||||||||||
|
Figure. GeneMedi's anti-2019-nCoV Spike Neutralizing antibodies (Nabs) block Recombinant 2019-nCoV(SARS-CoV-2) Spike RBD protein (GMP-V-2019nCoV-SRBD001) and hACE2 (GMP-H-ACE2002) binding. |
GeneMedi's efficacy evaluation solutions will be useful for your anti-COVID-19 candidates funtional assay and evaluation,which is including:
1) Types of Vaccines (by testing immunized serum from human, mouse, NHP etc.)
2) Neutralizing antibodies
3) Peptides blockers (peptide inhibitors)
4) Compounds targeting Spike induced cell-fusion.
INFORMATION COLLECTION
1. Landscape of global COVID-19 vaccine candidates development
| Platform | Type of candidate vaccine | Developer | Coronavirus target |
Current stage of clinical evaluation/regulatory status- Coronavirus candidate |
Same platform for non-Coronavirus candidates | |||||||||||
| DNA | DNA plasmids containing S-gene | Biosun Pharmed | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | DNA plasmid vaccine | Globe Biotech Limited, Bangladesh | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | Plasmid DNA, nanostructured RBD | National institute of Chemistry, Slovenia | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA |
DNA, engineered vaccine inserts compatible with multiple delivery systems |
DIOSynVax Ltd / University of Cambridge |
SARS-CoV-2 and Sarbeco-CoV |
Pre-Clinical | ||||||||||||
| DNA | DNA vaccine | Ege University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | DNA plasmid vaccine RBD&N | Scancell/University of Nottingham/ Nottingham Trent University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | DNA plasmid vaccine S,S1,S2,RBD &N | National Research Centre, Egypt | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | DNA with electroporation |
Karolinska Institute / Cobra Biologics (OPENCORONA Project) |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | DNA with electroporation | Chula Vaccine Research Center | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | DNA | Takis/Applied DNA Sciences/Evvivax | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | Plasmid DNA, Needle-Free Delivery | Immunomic Therapeutics, Inc./EpiVax, Inc./PharmaJet | SARS-CoV2 | Pre-Clinical | SARS | |||||||||||
| DNA | DNA vaccine | BioNet Asia | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | msDNA vaccine | Mediphage Bioceuticals/University of Waterloo | SARS-CoV2 | Pre-Clinical | ||||||||||||
| DNA | DNA vaccine | Entos Pharmaceuticals | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated | Inactivated + Alum | Shifa Pharmed | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated | Inactivated | Milad Pharmaceutics Co. | SARS-CoV2 | Pre-Clinical | MMR, IPV | |||||||||||
| Inactivated | Inactivated | Zista Kian Azma Co. | SARS-CoV2 | Pre-Clinical | MMR, IPV | |||||||||||
| Inactivated | Inactivated | Kocak Farma Ilac ve Kimya San. A.S. | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated |
Egg-based, inactivated, whole chimeric Newcastle Disease Virus (NDV) expressing membrane-anchored pre-fusion-stabilized trimeric SARS-CoV-2 S protein (Hexapro) + CpG 1018 |
Institute of Vaccines and Medical Biologicals (IVAC; Vietnam) / Dynavax / PATH |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated |
Egg-based, inactivated, whole chimeric Newcastle Disease Virus (NDV) expressing membrane-anchored pre-fusion-stabilized trimeric SARS-CoV-2 S protein (Hexapro) + CpG 1018 |
Government Pharmaceutical Organization (GPO; Thailand) / Dynavax / PATH | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated |
Egg-based, inactivated, whole chimeric Newcastle Disease Virus (NDV) expressing membrane-anchored pre-fusion-stabilized trimeric SARS-CoV-2 S protein (Hexapro) + CpG 1018 |
Institute Butantan (Brazil) / Dynavax / PATH | SARS-CoV-2 | Pre-clinical | ||||||||||||
| Inactivated | Inactivated + alum | KM Biologics | SARS-CoV2 | Pre-Clinical | JE, Zika | |||||||||||
| Inactivated | Inactivated | Selcuk University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated | Inactivated | Erciyes University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated | Inactivated whole virus | National Research Centre, Egypt | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated | Inactivated | SARS-CoV2 | Pre-Clinical | |||||||||||||
| Inactivated | TBD | Osaka University/ BIKEN/ NIBIOHN | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated | Inactivated + CpG 1018 | Sinovac/Dynavax | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Inactivated | Inactivated + CpG 1018 | Valneva/Dynavax | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Live Attenuated Virus | Codon deoptimized live attenuated vaccines |
Mehmet Ali Aydinlar University / Acıbadem Labmed Health Services A.S. |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| Live Attenuated Virus | Codon deoptimized live attenuated vaccines | Indian Immunologicals Ltd/Griffith University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Non Replicating Viral Vector | Ad 5 vector for intranasal administration | University of Helsinki & University of Eastern Finland | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Non Replicating Viral Vector | Adenovirus Type 5 Vector | Globe Biotech Limited, Bangladesh | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Non-Replicating Viral Vector | Sendai virus vector | ID Pharma | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Non-Replicating Viral Vector | Adenovirus-based | Ankara University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Non-Replicating Viral Vector | Adeno-associated virus vector (AAVCOVID) | Massachusetts Eye and Ear/Massachusetts General Hospital/AveXis | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Non-Replicating Viral Vector | MVA encoded VLP | GeoVax/BravoVax | SARS-CoV2 | Pre-Clinical | LASV, EBOV, MARV, HIV | |||||||||||
| Non-replicating viral vector | MVA-S encoded | DZIF – German Center for Infection Research/IDT Biologika GmbH | SARS-CoV2 | Pre-clinical | Many | |||||||||||
| Non-replicating viral vector | MVA-S | IDIBAPS-Hospital Clinic, Spain | SARS-CoV2 | Pre-clinical | ||||||||||||
| Non-Replicating Viral Vector | Intranasal Ad5 vaccine encoding RBD | Altimmune, Inc. | SARS-CoV2 | IND filed |
Influenza (NasoVAX), Anthrax (NasoShield) |
|||||||||||
| Non-Replicating Viral Vector | Adeno5-based | Erciyes University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Non-Replicating Viral Vector | Ad5 S (GREVAX™ platform) | Greffex | SARS-CoV2 | Pre-Clinical | MERS | |||||||||||
| Non-Replicating Viral Vector | Oral Ad5 S | Stabilitech Biopharma Ltd | SARS-CoV2 | Pre-Clinical | Zika, VZV, HSV-2 and Norovirus | |||||||||||
| Non-Replicating Viral Vector | adenovirus-based + HLA-matched peptides | Valo Therapeutics Ltd | Pan-Corona | Pre-Clinical | ||||||||||||
| Non-Replicating Viral Vector | Vaxart | SARS-CoV2 | Pre-Clinical | InfA, CHIKV, LASV, NORV; EBOV, RVF, HBV, VEE | ||||||||||||
| Non-Replicating Viral Vector | MVA expressing structural proteins | Centro Nacional Biotecnología (CNB-CSIC), Spain | SARS-CoV2 | Pre-Clinical | Multiple candidates | |||||||||||
| Non-Replicating Viral Vector |
parainfluenza virus 5 (PIV5)-based vaccine expressing the spike protein |
University of Georgia/University of Iowa | SARS-CoV2 | Pre-Clinical | MERS | |||||||||||
| Non-Replicating Viral Vector |
Recombinant deactivated rabies virus containing S1 |
Bharat Biotech/Thomas Jefferson University | SARS-CoV2 | Pre-Clinical | HeV, NiV, EBOV, LASSA, CCHFV, MERS | |||||||||||
| Non-Replicating Viral Vector | Influenza A H1N1 vector | National Research Centre, Egypt | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Non-Replicating Viral Vector | Newcastle disease virus expressing S | Icahn School of Medicine at Mount Sinai | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Recombinant spike with adjuvant | Iran | SARS-CoV2 | Pre-Clinical | Multiple candidates | |||||||||||
| Protein Subunit | Recombinant S protein produced in BEVS | Tampere University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
RBD protein delivered in mannose- conjugated chitosan nanoparticle |
Ohio State University / Kazakh National Agrarian University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
Recombinant spike protein with Essai O/W 1849101 adjuvant |
Kazakh National Agrarian University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Peptides | Neo7Logic | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
Recombinant spike protein with Essai O/W 1849101 adjuvant |
Kazakh National Agrarian University, Kazakhstan / National Scientific Center for Especially Dangerous Infections |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Recombinant S protein | Max-Planck-Institute of Colloids and Interfaces | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
RBD protein (baculovirus production) + FAR- Squalene adjuvant |
Farmacológicos Veterinarios SAC (FARVET SAC) / Universidad Peruana Cayetano Heredia (UPCH) |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Protein Subunit | Research Institute for Biological Safety Problems, Rep of Kazakhstan | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | RBD-protein | Mynvax | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Recombinant S protein | Izmir Biomedicine and Genome Center | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Peptide + novel adjuvant | Bogazici University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
S subunit intranasal liposomal formulation with GLA/3M052 adjs. |
University of Virginia | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
S-Protein (Subunit) + Adjuvant, E coli based Expression |
Helix Biogen Consult, Ogbomoso & Trinity Immonoefficient Laboratory, Ogbomoso, Oyo State, Nigeria. |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Protein Subunit S,N,M&S1 protein | National Research Centre, Egypt | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Protein Subunit | University of San Martin and CONICET, Argentina | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | RBD protein fused with Fc of IgG + Adj. | Chulalongkorn University/GPO, Thailand | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Capsid-like Particle | AdaptVac (PREVENT-nCoV consortium) | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
Drosophila S2 insect cell expression system VLPs |
ExpreS2ion | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Peptide antigens formulated in LNP | IMV Inc | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | S protein | WRAIR/USAMRIID | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | S protein +Adjuvant | National Institute of Infectious Disease, Japan/Shionogi/UMN Pharma | SARS-CoV2 | Pre-Clinical | Influenza | |||||||||||
| Protein Subunit | VLP-recombinant protein + Adjuvant |
Osaka University/ BIKEN/ National Institutes of Biomedical Innovation, Japan |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | microneedle arrays S1 subunit | Univ. of Pittsburgh | SARS-CoV2 | Pre-Clinical | MERS | |||||||||||
| Protein Subunit | Peptide | Vaxil Bio | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Peptide | Flow Pharma Inc | SARS-CoV2 | Pre-Clinical |
Ebola, Marburg, HIV, Zika, Influenza, HPV therapeutic vaccine, BreastCA vaccine |
|||||||||||
| Protein Subunit | S protein | AJ Vaccines | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Ii-Key peptide | Generex/EpiVax | SARS-CoV2 | Pre-Clinical | Influenza, HIV, SARS-CoV | |||||||||||
| Protein Subunit | S protein | EpiVax/Univ. of Georgia | SARS-CoV2 | Pre-Clinical | H7N9 | |||||||||||
| Protein Subunit | Protein Subunit EPV-CoV-19 | EpiVax | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | gp-96 backbone | Heat Biologics/Univ. Of Miami | SARS-CoV2 | Pre-Clinical | NSCLC, HIV, malaria, Zika | |||||||||||
| Protein Subunit | Subunit vaccine | FBRI SRC VB VECTOR, Rospotrebnadzor, Koltsovo | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | S1 or RBD protein | Baylor College of Medicine | SARS-CoV2 | Pre-Clinical | SARS | |||||||||||
| Protein Subunit | Subunit protein, plant produced | iBio/CC-Pharming | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
Recombinant protein, nanoparticles (based on S-protein and other epitopes) |
Saint-Petersburg scientific research institute of vaccines and serums | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
COVID-19 XWG-03 truncated S (spike) proteins |
Innovax/Xiamen Univ./GSK | SARS-CoV2 | Pre-Clinical | HPV | |||||||||||
| Protein Subunit | Adjuvanted microsphere peptide | VIDO-InterVac, University of Saskatchewan | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
Synthetic Long Peptide Vaccine candidate for S and M proteins |
OncoGen | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
Oral E. coli-based protein expression system of S and N proteins |
MIGAL Galilee Research Institute | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Nanoparticle vaccine | LakePharma, Inc. | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit |
Plant-based subunit (RBD-Fc + Adjuvant) |
Baiya Phytopharm/ Chula Vaccine Research Center | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | OMV-based vaccine | Quadram Institute Biosciences | SARS-CoV2 | Pre-Clinical | Flu A, plague | |||||||||||
| Protein Subunit | OMV-based vaccine | BiOMViS Srl/Univ. of Trento | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein subunit |
structurally modified spherical particles of the tobacco mosaic virus (TMV) |
Lomonosov Moscow State University | SARS-CoV2 | Pre-Clinical | rubella, rotavirus | |||||||||||
| Protein Subunit | Spike-based | University of Alberta | SARS-CoV2 | Pre-Clinical | Hepatitis C | |||||||||||
| Protein Subunit | Recombinant S1-Fc fusion protein | AnyGo Technology | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Recombinant protein | Yisheng Biopharma | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Recombinant S protein in IC-BEVS | Vabiotech | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Orally delivered, heat stable subunit | Applied Biotechnology Institute, Inc. | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Peptides derived from Spike protein | Axon Neuroscience SE | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Protein Subunit | MOGAM Institute for Biomedical Research, GC Pharma | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | RBD-based | Neovii/Tel Aviv University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Outer Membrane Vesicle (OMV)-subunit | Intravacc/Epivax | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Outer Membrane Vesicle(OMV)-peptide | Intravacc/Epivax | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Spike-based (epitope screening) | ImmunoPrecise/LiteVax BV | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Protein Subunit | Spiked-based |
Nanografi Nano Technology, Middle East Technical University, Ankara University, |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Bacteria Vector |
Oral Salmonella enteritidis (3934Vac) based protein expression system of RBD |
Farmacológicos Veterinarios SAC (FARVET SAC) / Universidad Peruana Cayetano Heredia (UPCH) |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector |
Intranasal Newcastle disease virus vector (rNDV-FARVET) expressing RBD |
Farmacológicos Veterinarios SAC (FARVET SAC) / Universidad Peruana Cayetano Heredia (UPCH) |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector | YF17D Vector | KU Leuven | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector | Measles Vector | Cadila Healthcare Limited | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector | Measles Vector | FBRI SRC VB VECTOR, Rospotrebnadzor, Koltsovo | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector | Measles Virus (S, N targets) | DZIF – German Center for Infection Research/CanVirex AG | SARS-CoV2 | Pre-clinical | Zika, H7N9, CHIKV | |||||||||||
| Replicating Viral Vector | Horsepox vector expressing S protein | Tonix Pharma/Southern Research | SARS-CoV2 | Pre-Clinical | Smallpox, monkeypox | |||||||||||
| Replicating Viral Vector | Live viral vectored vaccine based on attenuated influenza virus backbone (intranasal) | BiOCAD and IEM | SARS-CoV2 | Pre-Clinical | Influenza | |||||||||||
| Replicating Viral Vector | Recombinant vaccine based on Influenza A virus, for the prevention of COVID-19 (intranasal) | FBRI SRC VB VECTOR, Rospotrebnadzor, Koltsovo | SARS-CoV2 | Pre-Clinical | Influenza | |||||||||||
| Replicating Viral Vector |
Attenuated Influenza expressing an antigenic portion of the Spike protein |
Fundação Oswaldo Cruz and Instituto Buntantan | SARS-CoV2 | Pre-Clinical | Influenza | |||||||||||
| Replicating Viral Vector | Influenza vector expressing RBD | University of Hong Kong | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector | Replicating VSV vector-based DC-targeting | University of Manitoba | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector | VSV-S | University of Western Ontario | SARS-CoV2 | Pre-Clinical | HIV, MERS | |||||||||||
| Replicating Viral Vector | VSV-S | Aurobindo | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector | VSV vector | FBRI SRC VB VECTOR, Rospotrebnadzor, Koltsovo | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector |
M2-deficient single replication (M2SR) influenza vector |
UW–Madison/FluGen/Bharat Biotech | SARS-CoV2 | Pre-Clinical | influenza | |||||||||||
| Replicating Viral Vector |
Newcastle disease virus vector (NDV-SARS- CoV-2/Spike) |
Intravacc/ Wageningen Bioveterinary Research/Utrecht Univ. | SARS-CoV2 | Pre-Clinical | ||||||||||||
| Replicating Viral Vector | Avian paramyxovirus vector (APMV) | The Lancaster University, UK | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | mRNA | Providence Therapeutics | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | mRNA | Cell Tech Pharmed | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | mRNA | ReNAP Co. | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | D614G variant LNP-encapsulated mRNA | Globe Biotech Ltd | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | saRNA formulated in a NLC | Infectious Disease Research Institute/ Amyris, Inc. | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | LNP-encapsulated mRNA encoding S | Max-Planck-Institute of Colloids and Interfaces | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | Self-amplifying RNA | Gennova | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | mRNA | Selcuk University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | LNP-mRNA | Translate Bio/Sanofi Pasteur | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | LNP-mRNA | CanSino Biologics/Precision NanoSystems | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA |
LNP-encapsulated mRNA cocktail encoding VLP |
Fudan University/ Shanghai JiaoTong University/RNACure Biopharma | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | LNP-encapsulated mRNA encoding RBD | Fudan University/ Shanghai JiaoTong University/RNACure Biopharma | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA |
Replicating Defective SARS-CoV-2 derived RNAs |
Centro Nacional Biotecnología (CNB-CSIC), Spain | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | LNP-encapsulated mRNA | University of Tokyo/ Daiichi-Sankyo | SARS-CoV2 | Pre-Clinical | MERS | |||||||||||
| RNA | Liposome-encapsulated mRNA | BIOCAD | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | Several mRNA candidates | RNAimmune, Inc. | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | mRNA | FBRI SRC VB VECTOR, Rospotrebnadzor, Koltsovo | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | mRNA | China CDC/Tongji University/Stermina | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | LNP-mRNA | Chula Vaccine Research Center/University of Pennsylvania | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | mRNA in an intranasal delivery system | eTheRNA | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | mRNA | Greenlight Biosciences | SARS-CoV2 | Pre-Clinical | ||||||||||||
| RNA | mRNA | IDIBAPS-Hospital Clinic, Spain | SARS-CoV2 | Pre-Clinical | ||||||||||||
| T-cell based |
CD8 T cell peptide targeting (S, M, N) and (NSPs) SARS-CoV-2 proteins |
OSE immunotherapeutics | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | Plant derived VLP | Shiraz University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | VLPs produced in BEVS | Tampere University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | VLP | Max Planck Institute for Dynamics of Complex Technical Systems | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP |
Virus-like particle-based Dendritic Cell(DC)- targeting vaccine |
University of Manitoba | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | VLP | Bezmialem Vakif University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | VLP | Middle East Technical University | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | Enveloped Virus-Like Particle (eVLP) | VBI Vaccines Inc. |
SARS-CoV-2, SARS-CoV, MERS-CoV |
Pre-Clinical | CMV, GBM, Zika | |||||||||||
| VLP | S protein integrated in HIV VLPs |
IrsiCaixa AIDS Research/IRTA-CReSA/Barcelona Supercomputing Centre/Grifols |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | VLP + Adjuvant |
Mahidol University/ The Government Pharmaceutical Organization (GPO)/Siriraj Hospital |
SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP |
Virus-like particles, lentivirus and baculovirus vehicles |
Navarrabiomed, Oncoimmunology group | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP |
Virus-like particle, based on RBD displayed on virus-like particles |
Saiba GmbH | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | ADDomerTM multiepitope display | Imophoron Ltd and Bristol University’s Max Planck Centre | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | Unknown | Doherty Institute | SARS-CoV2 | Pre-Clinical | ||||||||||||
| VLP | VLP | OSIVAX |
SARS-CoV1 SARS-CoV2 |
Pre-Clinical | ||||||||||||
| VLP | eVLP | ARTES Biotechnology | SARS-CoV2 | Pre-Clinical | malaria | |||||||||||
| VLP | VLPs peptides/whole virus | Univ. of Sao Paulo | SARS-CoV2 | Pre-Clinical | ||||||||||||
2. COVID-19 Guidance and information collection on the new mutant variants of the SARS-CoV-2 (2019nCoV) virus
- COVID-19 (SARS-CoV-2): information about the new virus variant
- New SARS-COV-2 variant: information and risk assessment
- Central Alerting System (CAS) alert
- New variant clustering in households analysis (ONS)
- SARS-CoV-2 lateral flow antigen tests: evaluation of VUI-202012/01
- WTO: Statement of the WHO Working Group on COVID-19 Animal Models (WHO-COM) about the UK and South African SARS-CoV-2 new variants.
- EMA guidance for COVID-19 vaccine
- Investigation of novel SARS-CoV-2 variant: Variant of Concern 202012/01
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 6
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 5
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 4
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 3
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 2
• Investigation of novel SARS-CoV-2 variant: 202012/01. Technical briefing 1
New variant of SARS-COV-2 (2019nCOV) B.1.1.7 lineage spreaded in UK
The world is in midst of the COVID-19 pandemic. Recently a novel SARS-COV-2 (2019nCOV) lineage, the B.1.1.7 lineage, with serials of site mutation, shows stronger infection ability in the UK. The SARS-COV-2 B.1.1.7 lineage carries a larger than a usual number of coronavirus genetic changes.
Extended Reading: Preliminary genomic characterisation of an emergent SARS-CoV-2 lineage in the UK defined by a novel set of spike mutations
|
Relative products collection: GeneMedi products for New variant of SARS-COV-2 (2019nCOV) UK B.1.1.7 lineage |
New variant of SARS-COV-2 (2019nCOV) 501Y.V2 lineage(B.1.351) spreaded in South African SARS-CoV-2
The South African variant is characterized by eight lineage-defining mutations in the spike protein including three key residues in the receptor binding domain (K417N, E484K and N501Y) and is referred to as lineage 501Y.V2.
Extended Reading:
Alert Notification: New SARS-CoV-2 variant with multiple spike protein mutations
|
Relative products collection: GeneMedi products for New variant of SARS-COV-2 (2019nCOV) South Africa 501Y.V2 lineage(B.1.351) |
3. COVID-19 News and announcements collection on the new mutant variants of the SARS-CoV-2 (2019nCoV) virus
- PHE investigating a novel variant of COVID-19
- Rapid evaluation confirms lateral flow devices effective in detecting new COVID-19 variant
- Confirmed cases of COVID-19 variant from South Africa identified in UK
- Statement from Chief Medical Officer, Professor Chris Whitty, about new strain of COVID-19
- NERVTAG statements on COVID-19 (SARS-CoV-2)
• NERVTAG/SPI-M Extraordinary meetingon SARS-CoV-2 variant of concern 202012/01 (variant B.1.1.7)
• NERVTAG meetingon SARS-CoV-2 variant under investigation VUI-202012/01
• CPR as an AGP - Evidence review and NERVTAG consensus
4. About COVID-19 Pandemic and SARS-CoV-2 Vaccine
Coronavirus Disease 2019 (COVID-19) is a novel viral pneumonia caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). First discovered in Wuhan, a city in Hubei province of China, COVID-19 has already broken out throughout the world and posed a great threat to the public health, especially in Europe and North America now. Additionally, person-to-person transmission of COVID-19 disease is reported to be extremely rapid [158-160]. To date, more than one million cases were infected with COVID-19 and over 55,000 deaths occurred. Therefore, it is really urgent and noteworthy to develop the vaccines specific to COVID-19/SARS-CoV-2.
Belonging to the Betacoronavirus genus family, SARS-CoV-2 is 60~200nm in diameter and encapsidates a large positive-sense, single-stranded RNA virus (26-32kb) with many spikes on the virus capsid (Fig. 17A). The RNA genome of SARS-CoV-2 encodes several accessory proteins and structural proteins, such as nucleocapsid (N) protein, envelope (E) protein, membrane (M) protein, and spike (S) protein (Fig. 10B). Although the detailed mechanism of SARS-CoV-2 infection has not been clearly illuminated, several studies demonstrated that SARS-CoV-2 enters human cells via utilizing spike (S) protein to bind to the angiotensin converting enzyme (ACE2) on the surface of target cell [161, 162].
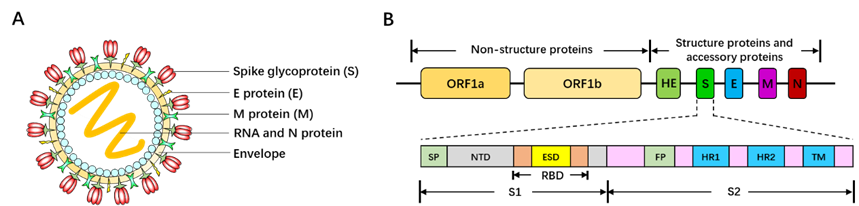
Figure S1. SARS-CoV-2 capsid structure and genome map. (A) Three-dimensional structure diagram of SARS-CoV-2. (B) Genome organization of SARS-CoV-2 [158]. ORF: open reading frame. E: envelope. M: membrane. N: nucleocapsid. HR1: heptad repeat 1. HR2: heptad repeat 2. SP: signal peptide. NTD: N-terminal domain. RBD: receptor binding domain. S: spike. S1: subunit 1. S2: subunit 2. TM: transmembrane domain.
Since the genome sequences of SARS-CoV were discovered and reported (https://www.gisaid.org/CoV2020/), a large number of pharmaceutical enterprises and research organizations are sparing all efforts to the vaccine development. Different companies utilize different targets and antigen epitopes. Some of the advances are listed in the following Table 10 (from WHO), and most of them focus on viral vector-based vaccines (replicating or non-replicating viral vector-based vaccines), recombinant protein (Spike), and nucleic acid-based vaccines. To date, two COVID-19 vaccines have entered Phase I clinical testing to assess the safety and potency of vaccines. One is mRNA-1273, was developed by Moderna Therapeutics, encoding a prefusion-stabilized form of Spike (S) protein [163] (https://www.nature.com/articles/d41587-020-00005-z). Another vaccine is recombinant protein of SARS-CoV-2 antigen, developed by Chinese Academy of Military Sciences, Institute of Military Medicine. It was predicted that these vaccines can be applied in clinics in a large scale as early as 2021 if they can successfully pass the clinical testing. Although there is a long way for theses vaccines to be applied for prevention and therapy of COVID-19, they indeed bring great hope and light to people all over the world.
References
1. 2 Korber, B. et al. Spike mutation pipeline reveals the emergence of a more transmissible form of SARS-CoV-2. bioRxiv Preprint., doi:10.1101/2020.04.29.069054 (2020). 2. Investigation of novel SARS-COV-2 variant. Public health England. 3. European Centre for Disease Prevention and Control. Rapid increase of a SARS-CoV-2 variant with multiple spike protein mutations observed in the United Kingdom – 20 December 2020.ECDC: Stockholm; 2020. 4. Li, G. & De Clercq, E. Therapeutic options for the 2019 novel coronavirus (2019-nCoV). Nature Reviews Drug Discovery, doi:10.1038/d41573-020-00016-0 (2020). 5. Haque, A. & Pant, A. B. Efforts at COVID-19 Vaccine Development: Challenges and Successes. Vaccines 8, doi:10.3390/vaccines8040739 (2020). 6. Dong, Y. et al. A systematic review of SARS-CoV-2 vaccine candidates. Signal Transduction and Targeted Therapy 5, doi:10.1038/s41392-020-00352-y (2020). 7. Andrew Rambaut1, N. L., Oliver Pybus, Wendy Barclay, Jeff Barrett5, Alesandro Carabelli6, Tom Connor, Tom Peacock, David L Robertson8, Erik Volz, . Preliminary genomic characterisation of an emergent SARS-CoV-2 lineage in the UK defined by a novel set of spike mutations. https://virological.org/ (2020). 8. Rapid increase of a SARS-CoV-2 variant with multiple spike protein mutations observed in the United Kingdom. European Centre for Disease Prevention and Control: Publications & data. 9. Zhang, L. et al. SARS-CoV-2 RNA reverse-transcribed and integrated into the human genome. bioRxiv Preprint. , doi:10.1101/2020.12.12.422516 (2020).






 PSV validated Data Poster Download
PSV validated Data Poster Download

