Protocol for virus injection in vivo
Animal experiment is essential in biomedical research. It contains establishment of a nude mouse tumor model and other disease model. Besides, gene transfection in vivo is included as well.
As previous reported, gene delivery targeting to specific organs can be achieved through AAV (Adeno-associated virus). But how to achieve virus injection in specific organs Three kinds of methods for AAV injection are fully described below:
Injection of AAV virus
basic experiment
virus transduction of targeted cell
Adeno-associated virus (AAV) is a advanced and safe tool for gene transfection in vivo. Compared with lentivirus and other viral vectors, AAV has a number of advantages as a gene transfer vector including high titer, low immunogenicity, long expression time, no pathogenicity and pathological toxicity, small spread of virus particles, multiple serotypes and relative tissue affinity.
For more information and protocol about Adeno-associated virus, see AAV vector system, AAV production, transduction and AAV gene therapy.
| Infection organ | Recommended serotype | Injection route | Animal | Injection volume (μl) |
| Heart | AAV9 | Multiple points in situ | Rat | 10-15/point, 3-5 points |
| Mouse | 10-15/point, 3-5 points | |||
| Tail vein | Rat | 250 (200g body weight) | ||
| Mouse | 100 | |||
| Liver | AAV8 or AAV9 | Tail vein | Rat | 200 (200g body weight) |
| Mouse | 100 | |||
| Whole brain | AAV-PHP.eB AAV-PHP.B | Tail vein | Rat | 200 (200g body weight) |
| Mouse | 100 | |||
| Lateral ventricle | AAV9 | Stereotactic | Rat | 1-5 |
| Mouse | 1-5 | |||
| Brain tissue | AAV9 | Stereotactic | Rat | 2-3 |
| Mouse | 1-2 | |||
| Fat | AAV9 | Intraperitoneal injection for intraperitoneal fat | Rat | 300 |
| Mouse | 150-200 | |||
| in situ injection for subcutaneous fat | Rat | 10-15/point, 3-5 points | ||
| Mouse | 10-15/point, 3-5 points | |||
| Skeletal muscle | AAV1 or AAV9 | In situ injection | Rat | 10-15/point, 3-5 points |
| Mouse | 10-15/point, 3-5 points | |||
| Eyes | AAV2, AAV10 or AAV-DJ | vitreous chamber injection | Rat | 3-5 |
| Mouse | 1-3 | |||
| subretinal space injection | Rat | 1-2 | ||
| Mouse | 1-2 | |||
| Lung | AAV6 | Intratracheal injection | Rat | 100 (200g weight) |
| Mouse | 50-75 | |||
| Kidney | AAV2 or AAV9 | Renal pelvis injection | Rat | 10-15/point, 3-5 points |
| Mouse | 10-15/point, 3-5 points | |||
| Intestine | AAV1 or AAV5 | Enema | Rat | 200 (200g weight) |
| Mouse | 100 |
Injection of tail vein
Infection sites: Heart, liver and whole brain (AAV-BBB)
1.Put mouse into the device for tail vein injection (If there is no such device, it can be retrofit with 50ml centrifuge tube. The whole device should be ventilated and fixed).
2.Disinfect the tail of mouse with 70% alcohol. Be careful to keep the tail warm, the blood vessels will contract if it's too cold.
3.Hold the mouse's tail with the thumb of the left forefinger, straighten it gently, then move the forefinger forward, bending the tail slightly. You can insert needle in the bending place and inject AAV (27-30G insulin needle, 100ul/mouse).
4.Inject slowly, Hold for 5-10 seconds to prevent virus from flowing back.
5. Pull out the needle, then press the injection site with fingers or dry aseptic cotton balls or gauze for a few seconds.
Hepatic portal vein injection (local delivery of AAV in liver)
Infection sites: liver
1.Inject Xylazin/Ketamine mixture (10mg/kg,100 mg/kg) (Xylazin/Ketamine, Sigma-Aldrich X1251, K2753) subcutaneous to anesthetize mouse.
2.Apply ointment on mouse's eyes (Oculentum simplex, Teva Pharmachemie) to prevent eyes from drying.
3.Adjust the mouse's abdomen to make it upward, shave the hair between the second ribs and the area between the fourth nipples. This step can be operated 1 days ahead without anesthesia.
4.Disinfect the depilation site repeatedly. Then cut 3cm from the ribs to the area of the fourth nipple with a scalpel. Be careful not to hurt other tissues such as mammary glands, intestines, liver and so on.
5.Carefully pull out the internal organs such as the large intestine and small intestine with a sterile cotton swab. And gently cover them with gauze to avoid touching, until the hepatic portal vein is seen.
6.Suck the virus to a 32g syringe in advance, insert the needle slowly into the hepatic portal vein at an angle of less than 5 degrees at a distance of about 1 cm below the liver. Insert 3-5 mm along the hepatic portal vein (Schematic diagram of the portal vein, if it is difficult to see, can be identified by anatomical mirror).
7.Inject the virus slowly. After the injection is completed, stay for 3-5s and then dispense the needle, and gently press it with a cotton swab for a while. Then cover the hepatic portal vein with hemostatic cotton, and gently press with a cotton swab for 5 minutes to help stop bleeding. Make sure hemostasis is done, otherwise continue to press the wound.
8.Then remove the hemostatic cotton (If the tissue is tightly attached to the hemostatic cotton, it can be moistened with sterile PBS).
9.Then gently put the organ back into the belly of the mouse. The wound sutured using a 4-0 line, approximately 10-15 needles.
10.100μl of bupivacaine (5 mg/ml) is given to the wound site to relieve pain. Hydration was performed by subcutaneous injection of 500 μl of sterile PBS with a 26G insulin needle. The entire operation took about 30 minutes.
11.After the experiment, it is necessary to keep the body of the mouse warm enough (such as a 37-degree heating lamp, a thermostat, etc.) to facilitate recovery.
Brain stereotactic positioning
Infected site: brain
1.Inject Xylazin/Ketamine mixture (10mg/kg,100 mg/kg) (Xylazin/Ketamine,Sigma-Aldrich X1251, K2753) subcutaneous to anesthetize mouse.
2.Shave the hair between the mouse's head and the ear, then place it on the warm tray of the stereo positioner.
3.Make the graduation between teeth and ears to zero. Apply ointment on mouse's eyes (Oculentum simplex, Teva Pharmachemie) to prevent eyes from drying.
4.Fix the upper incisors of the mouse into the slots of the fixed plate to ensure that the head remains fixed. Adjust Bregma and Lambma on the same sagittal line and horizontal plane. Make sure the mouse's nose is centered and stable.
5.Use a scalpel to peel off the skin of the mouse's head and scrape off the surface dura mater. If there is blood, please dry it with a cotton swab to ensure that the position of the bregma and lambda is clearly visible (plus schematic).
6.The bregma coordinates are homed to 0 (X, Y, Z = 0), ensuring that the lambda position is on the same level as the bregma.
7.Set the coordinates according to the target brain area. Once the coordinates are fixed, a marker was used to mark the skull, A hole of about 0.015 mm is drilled at the mark within 10 seconds. Be careful to prevent brain tissue from being damaged.
8.Suck the AAV into the syringe and inserted according to the coordinates set before. The virus is slowly injected at a rate of 0.05-0.1μl/min after inserting needle. Typically, each mouse is injected at a volume between 0.05μl and 2μl.
9.After the injection is completed, keep it for 10 minutes, then slowly return the needle.
10.If there is blood flowing out during the operation, use a cotton swab to dry the blood in time.
11.The last step is to suture the scalp. After the experiment, it is necessary to keep the body of the mouse warm enough (such as a 37-degree heating lamp, a thermostat, etc.) to facilitate recovery.
Intrathecal injection
Infected site: spinal nerve
1.Inject Xylazin/Ketamine mixture (10mg/kg,100 mg/kg) (Xylazin/Ketamine, Sigma-Aldrich X1251, K2753) subcutaneous to anesthetize mouse.
2.Apply ointment on mouse's eyes (Oculentum simplex, Teva Pharmachemie) to prevent eyes from drying.
3.Remove the hair at about 2 cm2 of the skin near the tail, in order to see where the needle is inserted.
4.Put the mouse on a special rack and place a 15ml centrifuge in the lower part of the body to arch the needle insertion site slightly.
5.Suck the virus into 25 micro Hamilton syringe with 30G needles.
6.Position the L6 (the most prominent one) of the spine and gently press it with your fingers to make it gentle.
7.Carefully insert the needle into the groove between the L5 vertebrae and L6 vertebra. Note that the tail of the mouse is slightly upturned, which indicates that the needle is successfully inserted into the myelin sheath.
8.Once successfully inserted, secure the needle with one hand and slowly inject 5-10μl of virus with the other hand. Note that the injection volume should be appropriate, small volume will increase the experimental error and a large volume will increase the intrathecal pressure.
9.If necessary, repeat the injection after 24h.
10.After the experiment, it is necessary to keep the body of the mouse warm enough (such as a 37-degree heating lamp, a thermostat, etc.) to facilitate recovery.
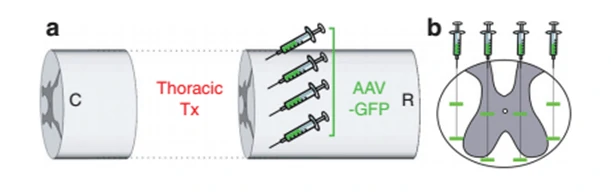
11.As previous reported, take 1μl as a point, Two cross-sections at different depths, 4 points on a cross-sectional line, a total of 8 points will be better, the schematic is as follows:
AAV(Adeno-Associated Virus) vector system
AAV Rep-Cap plasmids (serotypes-specific AAV RC plasmids)






